.png)
Most small food manufacturers do not fail food safety audits because they ignore food safety. They fail because their system does not match how their operation actually runs.
In many facilities, one QA person manages production, suppliers, documentation, and audit preparation. Documents are spread across folders, email threads, and spreadsheets. Everything exists in some form, but not in one place, not in one format, and not in a way that holds under audit pressure.
When the auditor arrives, they do not evaluate effort. They evaluate evidence. And if that evidence is not immediately accessible, consistent, and verifiable, the outcome is predictable.
This is why many teams feel surprised by audit results. From their perspective, the work has been done. From the auditor’s perspective, the system is not controlled.

The gap is not effort. It is structured. Most teams operate with fragmented systems that were never designed to support audit-level validation. Over time, inconsistencies accumulate. Documents are updated in one place but not another. Logs are completed but not reviewed. Supplier approvals exist but are not linked to incoming materials.
These gaps remain invisible during daily operations but become critical during audits. Auditors do not evaluate isolated documents. They assess whether the system behaves consistently over time and whether controls are applied in practice.
This is the structural reason audits fail.
What Food Safety Auditors Actually Look For
Most teams approach audits as if they are facility inspections. The preparation focuses on cleaning, organization, and GMP basics.
Those elements matter, but they are not what determines the result. Auditors start with documentation. The principle is simple. If it is not documented, it did not happen.
Standards such as SQF and BRCGS are designed as system audits, and many food manufacturers are now looking to automate those, especially with AI. The objective is to understand whether the operation is defined, whether it is followed consistently, and whether it can be proven through records.
This is why the first requests are always predictable. HACCP plan, CCP monitoring logs, corrective actions, supplier documentation, and traceability records.
Production is observed later. Documentation defines control.
The U.S. Food and Drug Administration repeatedly highlights missing or incomplete records as a key issue in enforcement actions, which can be seen across real cases in the
FDA warning letters database.
Auditors do not look for isolated mistakes. They look for patterns. Repeated gaps in records, identical values across logs, or the absence of deviations signal that the system is not functioning as intended.

Traceability is another critical dimension. A system that cannot connect suppliers, batches, and finished products within a defined timeframe is considered unreliable.
What matters is not whether the facility appears compliant on a given day, but whether the system demonstrates control across time.
The 5 Most Common Reasons Small Food Manufacturers Fail Food Safety Audits
These patterns appear consistently across audits. They are not tied to a specific product or certification. They reflect structural limitations in how systems are built and maintained.
#1 Documents Exist, But Cannot Be Found In Two Minutes
The fastest way to lose control during an audit is not the absence of a document, but the inability to retrieve it. According to ECFR, food safety plans must be reanalyzed at least once every 3 years or whenever significant changes occur.
When a record cannot be presented immediately, the perception shifts. The auditor is no longer evaluating the content of the document but the reliability of the system. This typically results from fragmentation. Documents live in different environments, and there is no single structure connecting them.

As operations grow, the problem compounds. More products, more batches, and more suppliers increase the volume of records. Without structure, retrieval becomes slower and less reliable.
This is where systems such as food manufacturing software solutions change the nature of the problem. Instead of storing documents, they structure relationships. HACCP plans connect to monitoring logs. Supplier documents connect to materials. Corrective actions connect to deviations.
The system becomes navigable rather than searchable. That distinction is what auditors interpret as control.
#2 The HACCP Plan Has Not Been Reviewed In Over A Year
HACCP plans often remain static while operations evolve. New ingredients, process adjustments, and supplier changes introduce new risks. When the plan does not reflect these changes, it becomes disconnected from reality.
Auditors detect this through comparison. The documented flow is matched against actual production. Any inconsistency signals that the system is not maintained. Food safety audit data across GFSI-recognized schemes show that a large share of non-conformities is due to systems that are not maintained, not to missing systems.

A static HACCP plan creates both compliance risk and operational risk. Controls may no longer address real hazards, and critical limits may not be appropriate.
Food Safety Platforms like IONI approach this differently by structuring HACCP plans around actual SOPs and maintaining continuous alignment. Changes in inputs or processes are reflected in the system without relying on periodic manual updates.
This dynamic structure is part of how FSMA, BRCGS, SQF compliance overview
translates into real operational control.
#3 Supplier COAs Are Expired Or Missing
Supplier control is one of the most consistent sources of audit findings. The issue is rarely the absence of documents. It is the lack of connection between them. COAs are stored separately from specifications. Approvals are tracked independently from incoming materials. This fragmentation creates blind spots.
As supplier networks grow, manual tracking becomes unreliable. Expiration dates are missed. Specifications are not validated consistently. Non-compliant materials can enter production.

This is why supplier-related non-conformities remain common across audits.
Systems like ingredients intelligence for food companies address this by linking supplier data, specifications, and batch validation. Incoming materials are automatically checked against requirements. Missing or non-compliant documentation is identified before use.
This shifts supplier control from periodic review to continuous validation.
#4 Corrective Actions Are Closed On Paper - Not Verified In Practice
Corrective actions often exist as completed forms rather than validated processes.
From a documentation perspective, the issue appears resolved. From a system perspective, there is no evidence that the solution worked.
Auditors look for a chain of logic. Root cause, corrective action, and verification. When any part is missing, the action is considered incomplete.

This is where minor issues escalate. Lack of verification signals that the system does not prevent recurrence.
IONI structures corrective actions as part of the operational flow. Deviations trigger actions. Root cause analysis is guided. Verification is required before closure.
This creates a feedback loop where issues lead to measurable improvements rather than isolated records.
#5 The Team Knows Procedures But Cannot Explain Them
Auditors assess understanding, not just documentation. When operators cannot explain procedures, it indicates that the system is not embedded in daily work.
This often results from the centralization of knowledge. One person understands the system, while others follow instructions without context.

Such systems are fragile. They depend on individuals rather than processes.
IONI distributes knowledge through workflow design. Operators interact with the system during routine tasks. Logging, validation, and corrective actions are guided in real time.
This reduces reliance on memory and aligns execution with documented procedures.
Over time, consistency improves because the system reinforces correct behavior continuously.
Your Food Safety Audit Checklist: What To Verify Before The Auditor Arrives
Audit readiness is not defined by how much documentation exists, but by how quickly and consistently the system can prove control across all areas of the operation.
In practice, auditors move through the system in layers. They start with documentation, validate HACCP logic, test supplier control, review monitoring records, and then examine how issues are handled and whether the system is reviewed internally. Each layer builds on the previous one.
Looking at audit outcomes across SQF and BRCGS, the same pattern appears repeatedly. Failures are rarely isolated to a single missing document. They occur when multiple elements are slightly misaligned. A checklist only works when it reflects that system logic.
Documentation
Documentation should be centralized, current, and consistent across all formats.
- All policies, SOPs, and records exist in a single structured system
- Version control is clear, with no duplicates or outdated documents in circulation
- Documents can be retrieved within seconds under pressure
- Formats are consistent across departments and processes
A common failure point is not missing documents, but delayed retrieval or conflicting versions, which signals a lack of control.
HACCP Plan
The HACCP plan must reflect actual operations, not historical assumptions.
- Process flow diagrams match real production steps
- Hazards are identified based on current ingredients and processes
- Critical control points and limits are clearly defined and relevant
- The plan has been reviewed and updated after any operational change
Across GFSI audits, one of the most consistent issues is drift between documented HACCP plans and real production, which undermines the credibility of the entire system.
Supplier Records
Supplier control must be continuous and connected to incoming materials.
- The approved supplier list is current and aligned with purchasing
- Certificates of analysis are valid, complete, and linked to batches
- Specifications are defined and consistently applied
- Expiration and renewal of supplier documentation are actively tracked
Fragmented supplier documentation is one of the most common sources of non-conformance because it breaks traceability and validation.
CCP Monitoring Logs
Monitoring records must demonstrate real control over time.
- Logs are complete, consistent, and recorded at required frequencies
- Data reflects realistic variation, not repeated identical values
- Deviations are documented when they occur
- Responses to deviations are recorded and linked to corrective actions
Auditors review logs across extended periods to identify patterns. Consistency and realism matter more than perfection.
Corrective Actions
Corrective actions must show that the system learns and improves.
- Root cause analysis is clearly documented
- Corrective actions are specific and relevant to the issue
- Evidence shows that the action was implemented
- Verification confirms that the issue has been resolved
Without verification, corrective actions remain procedural and do not demonstrate system effectiveness.
Internal Audit Records
Internal audits should demonstrate active system oversight.
- Audits are conducted regularly and cover all system areas
- Findings are documented with clear corrective actions
- Follow-up actions are tracked to completion
- Trends and recurring issues are identified and addressed
Internal audits are one of the strongest indicators auditors use to assess whether a system is maintained proactively or only prepared before certification.
When these elements are aligned, audit readiness becomes a natural outcome of daily operations rather than a last-minute effort. The structure behind the SQF certification in 90 days plan reflects how these components come together when systems are built progressively and maintained continuously.

For teams that want a practical version of this structure, the idea of a standardized SQF-ready checklist naturally emerges at this point as a way to validate system completeness before an audit scenario.
Already have SOPs or a HACCP plan? Upload your documents and see your SQF system built in one session inside IONI food manufacturing software solutions.
How To Build an Audit-Ready System Without a Full-Time Food Safety Team
This is the operating reality for most small food manufacturers. One QA person is responsible for production oversight, supplier coordination, documentation, and audit preparation. The system is expected to meet SQF or BRCGS requirements, but the resources behind it are limited.
The result is not a lack of knowledge. It is a lack of structure.
Where The Gap Comes From
The gap is structural, not procedural. Maintaining connections between HACCP, monitoring, suppliers, and corrective actions manually is not scalable. As operations grow, complexity increases, and consistency breaks down.
Audit failures happen when these connections cannot be demonstrated quickly and reliably.
How IONI Helps
IONI`s Food Safety System removes the need to build the system manually. You upload your SOPs, HACCP plans, specifications, and supplier documents.
a) The platform reads them and creates a structured system automatically.
- HACCP plans are mapped to real process steps
- CCPs are linked to monitoring requirements
- Documents are structured into a single system with clear relationships
Instead of static files, you get a system that reflects how your operation actually runs.
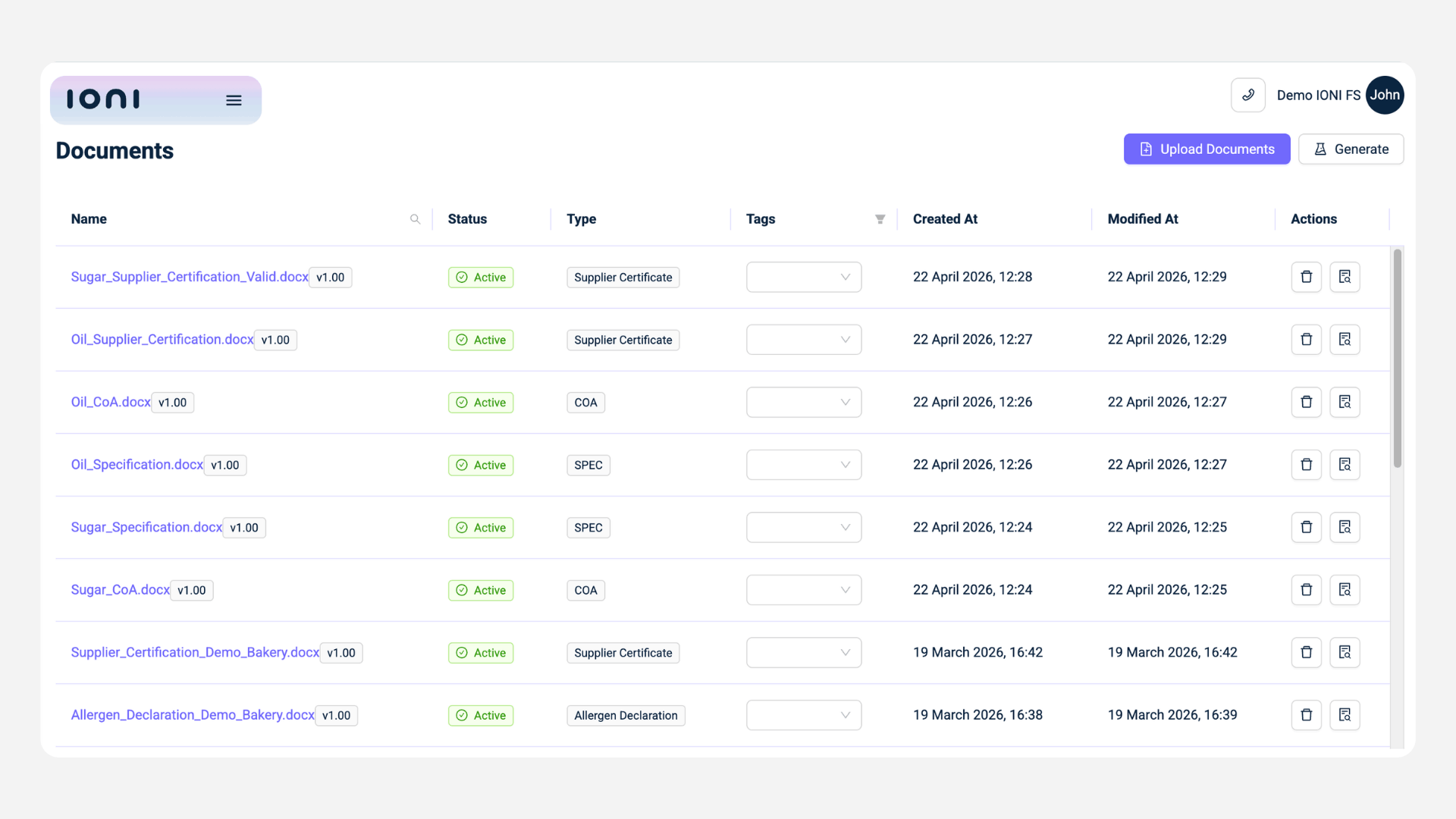
b) Once the structure exists, daily work becomes part of the system.
- Operators log CCP checks directly
- Entries are validated in real time against limits
- Deviations are detected immediately
- Corrective actions are created and tracked for verification
This ensures that records are not only complete but also meaningful and audit-ready.
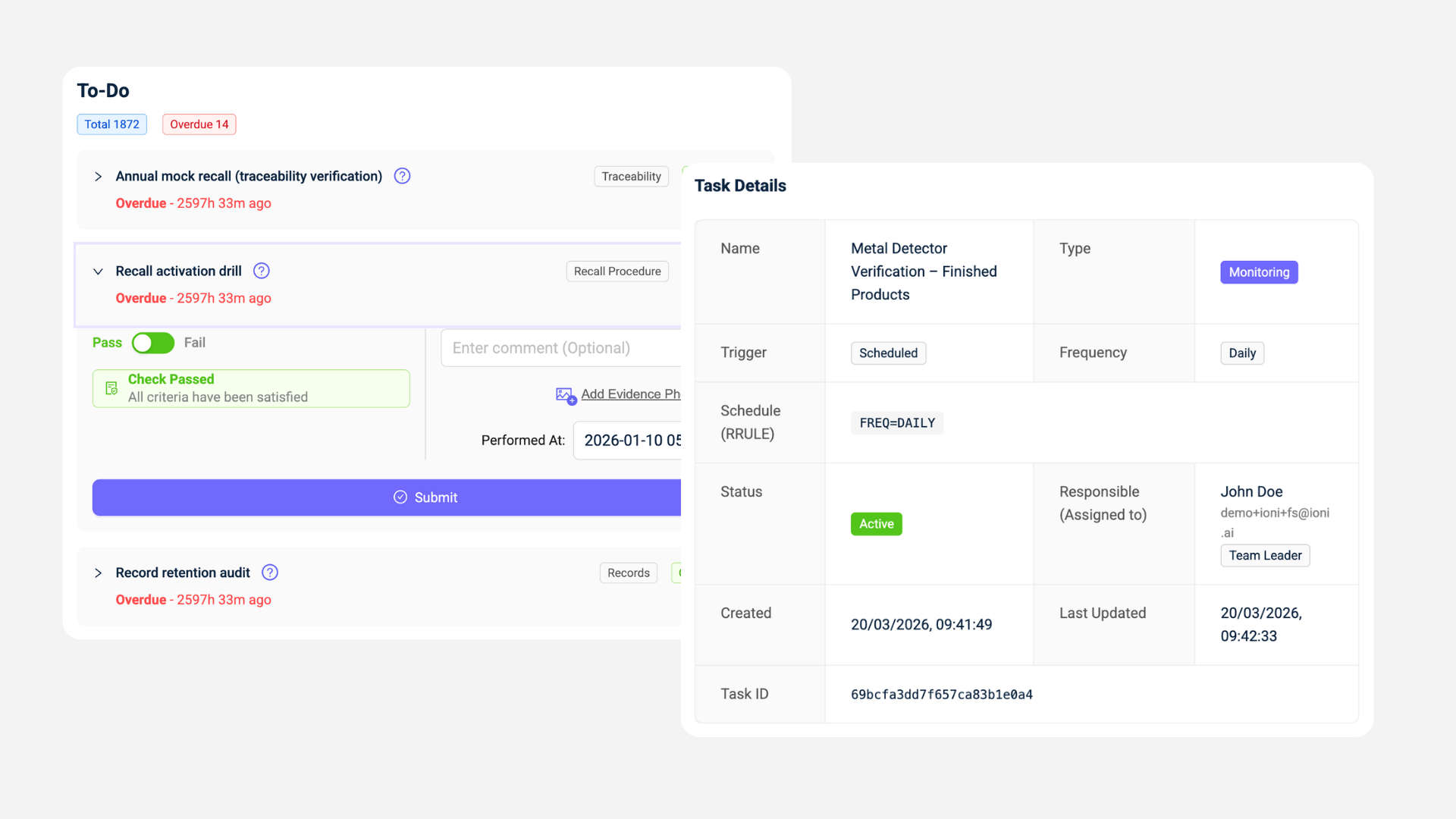
c) Supplier management becomes continuous instead of manual.
- COAs are checked automatically against specifications
- Missing or expired documents are flagged early
- Supplier approvals are linked to incoming materials
With ingredients intelligence for food companies, supplier control becomes part of the system, not a separate process.
.png)
d) Moreover, compliance does not stay static. Requirements change, processes evolve, and risks shift. IONI integrates AI regulatory intelligence to monitor regulatory updates and map them to your system.
This ensures that your documentation and processes remain aligned without requiring manual tracking.
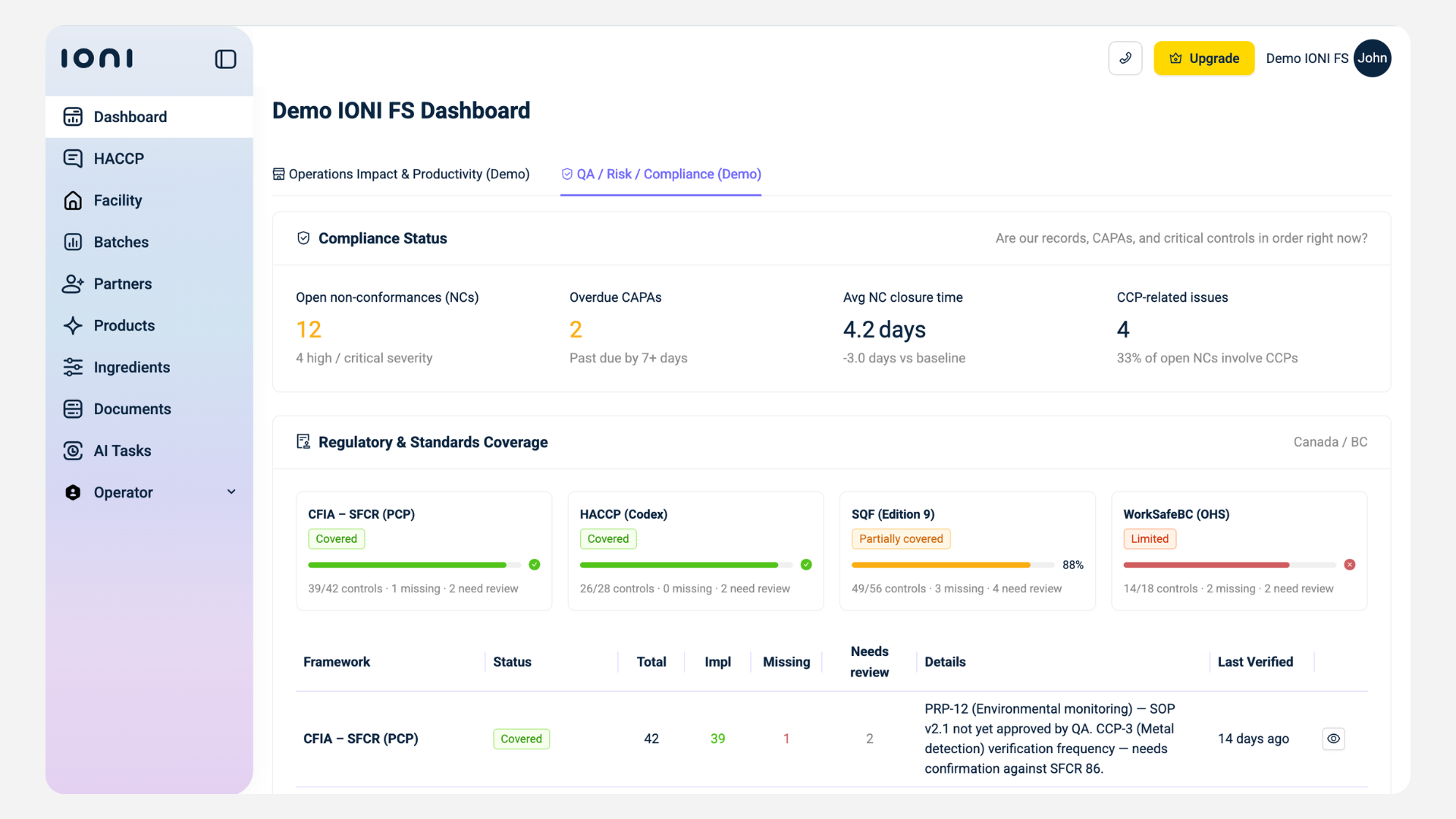
What Changes In Practice
The difference is operational.
- From documents stored across tools to a single connected system
- From periodic review to continuous validation
- From manual audit preparation to ongoing audit readiness
When an auditor asks for a record, it is retrieved instantly because it is already part of the system. When they assess control, they see not just procedures, but data proving consistent execution.
See your HACCP, suppliers, and audit records connected into one system in minutes, not weeks, inside IONI food manufacturing software solutions.
SQF Audit Checklist Vs BRC Audit Checklist: Key Differences For Small Manufacturers
For most small manufacturers, the choice between SQF and BRCGS is not just about certification. It defines how the system will be evaluated during audits and where weaknesses are most likely to surface.
Both standards are GFSI-recognized and require similar foundations, including HACCP, traceability, supplier control, and documented procedures. The difference lies in emphasis. SQF is more structured around system design and HACCP alignment, while BRCGS places additional weight on operational execution, culture, and consistency across teams.
Understanding this distinction helps explain why some audits fail even when documentation appears complete. A system that looks structured on paper may not hold under BRCGS if execution is inconsistent. A system that runs well operationally may still fail SQF if the structure and documentation are weak.
The comparison below highlights where these differences appear in practice.
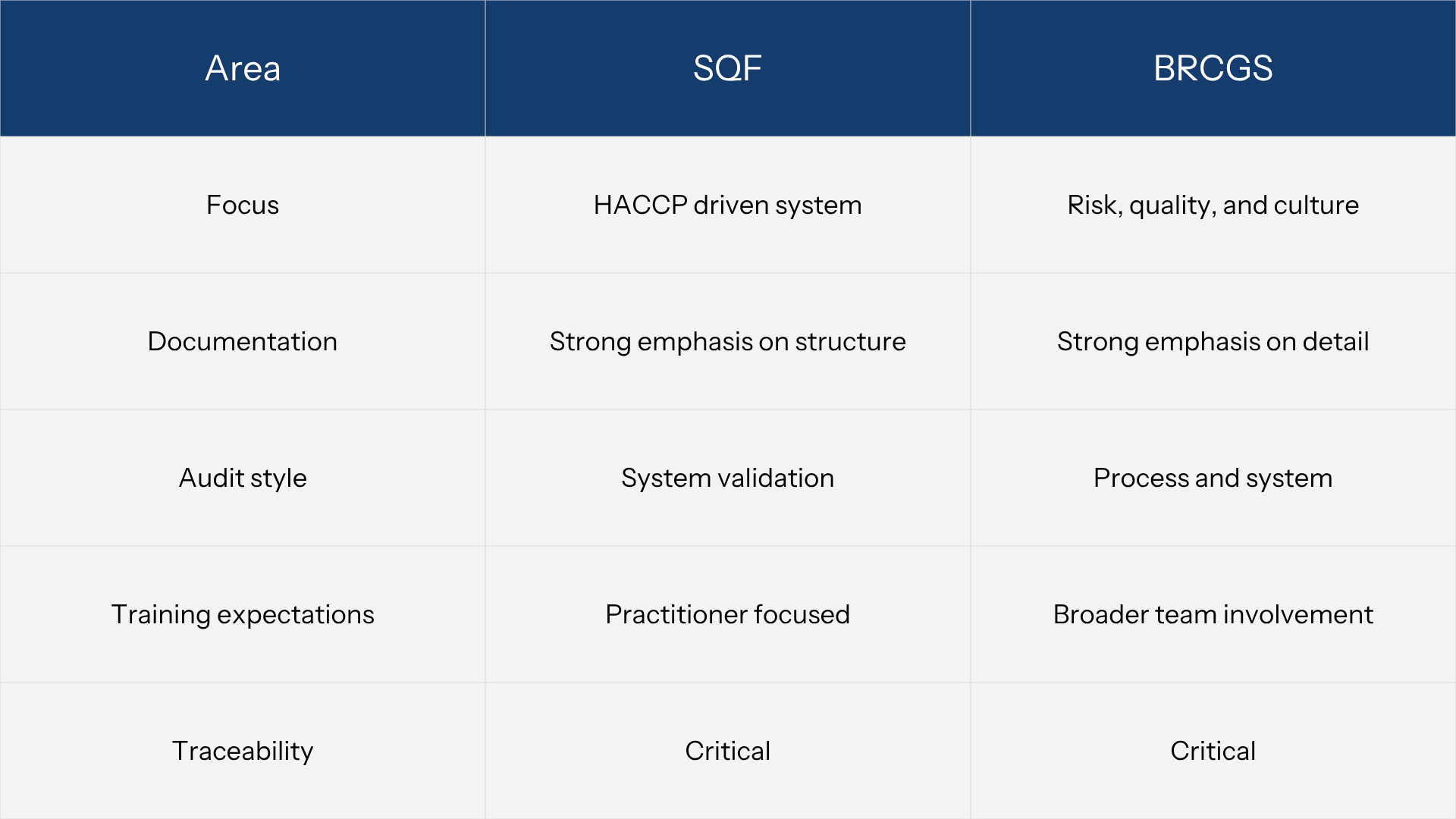
Both SQF and BRCGS ultimately assess the same foundation. Consistent records, validated controls, and clear corrective actions. The difference is how they expose weaknesses. SQF tends to reveal gaps in system structure, especially when HACCP plans, documentation, and monitoring are not properly connected.
BRCGS goes deeper into execution, highlighting whether procedures are actually embedded in daily operations and understood across the team. For small manufacturers, this means that even if documentation exists, it must function as a system that reflects real production, not just a set of files prepared for audit.
From Audit Preparation To Continuous Compliance
Most food manufacturers still operate in a preparation cycle. Documentation is reviewed before audits, gaps are identified, and fixes are implemented under time pressure.
This approach works in the short term, but it creates inconsistency. Issues are discovered late, corrective actions are reactive, and the system depends heavily on manual coordination.
A continuous compliance model changes that dynamic.
Instead of preparing for audits, the system validates itself during daily operations. Monitoring records are checked in real time. Deviations are identified immediately. Corrective actions are triggered and tracked as part of the process, not after the fact.
This reduces the risk of audit findings and improves operational control at the same time.The shift is already visible across the industry. As described in how AI is transforming food manufacturing in 2026 and the role of AI in modern SFCR compliance, AI systems are moving from isolated use cases into core compliance workflows.
IONI applies this model by connecting HACCP, monitoring, supplier control, and corrective actions into a single system that continuously validates.
Watch How IONI Food System Works
Instead of asking whether the system is ready for an audit, the question becomes whether the system is functioning correctly at any given moment.
Conclusion
Most audit failures are not caused by a lack of effort. They are caused by systems that are disconnected, static, and difficult to maintain.
Documentation exists, but it is not structured. Monitoring is performed, but it is not validated. Supplier data is collected, but it is not connected. Corrective actions are recorded, but not verified.
IONI resolves this by turning documentation into a working system. HACCP plans are built from real operations. Monitoring is validated in real time. Supplier data is checked continuously. Corrective actions are tracked through to verification.
Everything is connected.
When an auditor requests evidence, it is no longer a search across folders and emails. It is an immediate retrieval from a system that already reflects how the operation runs.
Bring your food safety plans, supplier documentation, and audit evidence together in a single structured system with IONI food manufacturing software solutions.
FAQ
What Does A Food Safety Auditor Check First
Auditors begin with documentation such as HACCP plans, monitoring logs, corrective actions, and supplier records. These provide an initial view of how structured and consistent the system is.
What Is The Most Common Reason For Failing An SQF Audit
The most common issue is inconsistency. Records may exist, but they are incomplete, outdated, or not aligned with actual operations. Corrective actions are another frequent gap, especially when they lack root cause analysis and verification.
How Often Should Internal Audits Be Conducted
At least once per year, and more frequently when processes change. Regular internal audits help identify gaps early and maintain system alignment.
Can The Same Checklist Be Used For SQF And BRCGS
There is overlap, but each standard emphasizes different aspects. SQF relatively focuses more on system structure, while BRCGS tends to place additional emphasis on execution and culture. Effective systems account for both.
What Happens If A Company Fails A Food Safety Audit
The company receives non-conformities that must be corrected within a defined timeframe. Depending on severity, certification may be delayed or denied until corrective actions are verified.
How To Keep Documentation Audit Ready Year-Round
Audit readiness is achieved through continuous validation rather than periodic preparation. When documentation, monitoring, supplier control, and corrective actions are connected and maintained in real time, the system remains ready without additional effort before audits.




.png)




