.png)
You just got the email.
Whole Foods approved your product. Costco wants to move forward. Walmart is ready to onboard you. One condition: SQF certification before you can start shipping.
Most manufacturers spend the first week researching. That week costs more than it seems.
SQFI itself recommends having at least 90 days of records available before the site audit. That makes the first week operationally important: if CCP logs, sanitation records, supplier checks, and corrective actions are not being collected yet, the certification timeline is already slipping.

This guide covers exactly what to do in the first 30 days: in the sequence that actually matters for small manufacturers without a dedicated food safety team.
Why Retailers Require SQF, and What They're Actually Asking For
When a major retailer requires SQF certification, they are requiring independent third-party proof that your food safety management system meets a globally recognized standard. Not a completed form. A functioning system - built, operated, then verified by an independent auditor.

SQF (Safe Quality Food) is a GFSI-benchmarked certification program administered by the SQF Institute. GFSI (Global Food Safety Initiative) is the benchmark framework that major retailers use to evaluate supplier food safety programs.
This aligns with the direction of US food regulation. Under FDA’s Preventive Controls for Human Food rule, covered facilities must have a written food safety plan that includes hazard analysis and risk-based preventive controls. FDA lists the small business compliance date as September 18, 2017, so for most established manufacturers, this is not a future requirement - it is already the baseline regulatory expectation.
All major US retailers require GFSI-recognized certification, and SQF is the most common standard in North America.
Here is what each major retailer requires:
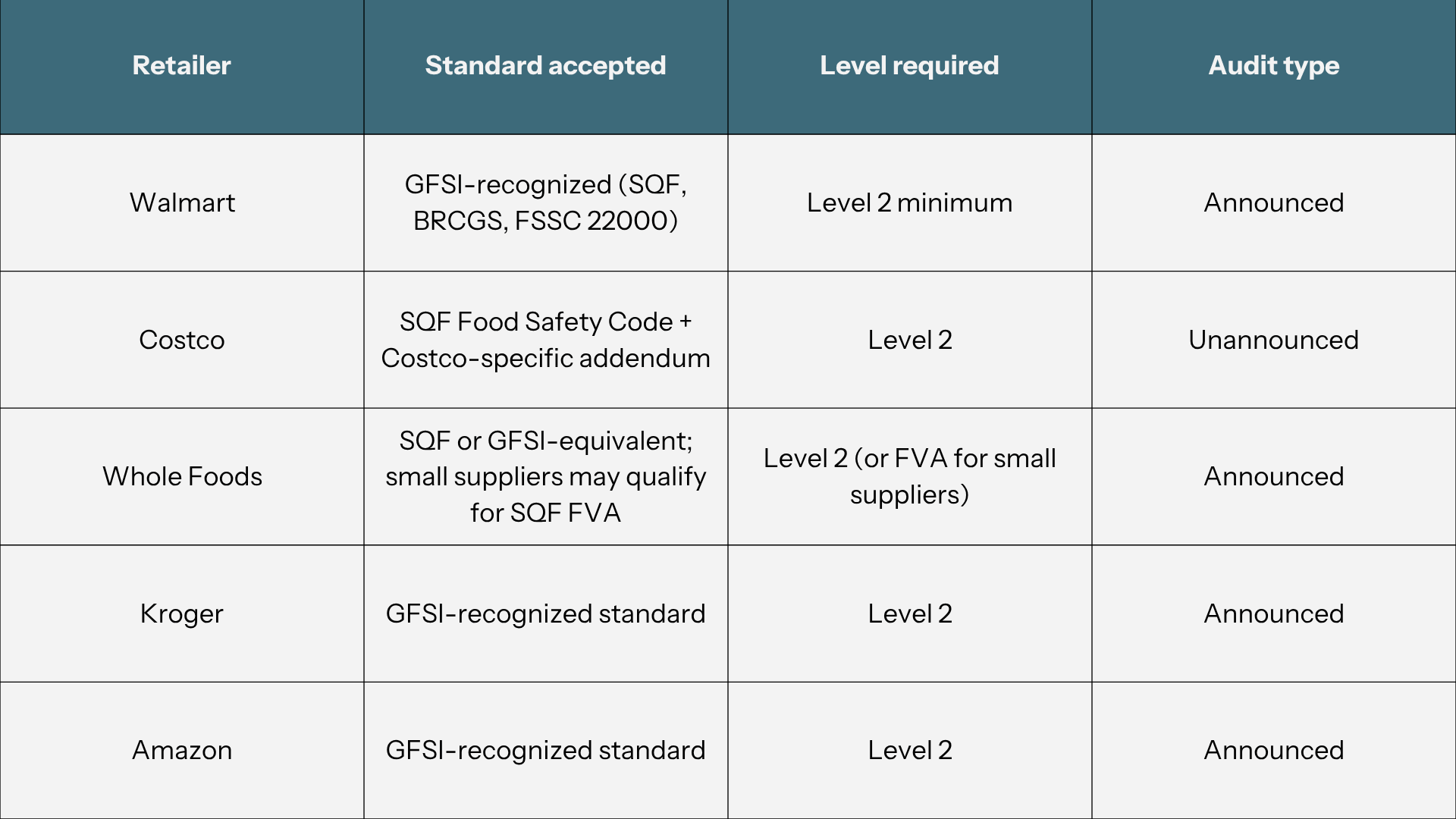
Costco note: Costco requires unannounced audits and has a facility-specific addendum that gets layered on top of the standard SQF audit. Confirm the current addendum version with your Costco buyer contact and at sqfi.com before scheduling anything.
Whole Foods note: Very small suppliers may be eligible for an SQF Fundamentals Verification Assessment (FVA) - a remote verification rather than a full on-site certification audit. SQFI states that FVA is 60% less than the cost of a typical onsite audit. It is not a full GFSI certification and does not satisfy requirements where GFSI-benchmarked Level 2 is required. Ask your Whole Foods contact whether FVA is accepted for your specific category before investing in full Level 2 preparation.
The single most important first step: confirm exactly which level and audit format your retailer requires.
For deeper context on how GFSI schemes differ, see: What Are GFSI Schemes And How To Choose The Right One
What SQF Certification Actually Involves for a Small Manufacturer
SQF certification is not a test. It is a two-stage audit of a management system you build and operate over time.
Stage 1. Desk audit: Your auditor reviews documentation remotely - your HACCP plan, SQF System Manual, procedures, and sample records. Incomplete or inconsistent documentation at this stage stops the process before a site visit happens.
Stage 2. Site audit: An auditor spends one to two days at your facility. They verify that your documentation matches how you actually operate, review monitoring logs, interview floor staff, and trace a production batch through your full process.
Between Stage 1 and Stage 2, you need a minimum of 90 days of operational records - CCP monitoring logs, corrective actions, supplier documentation, and internal audit results. This is how auditors verify that your system works under normal conditions, not just during audit prep.

As we may see, for a small manufacturer with an existing HACCP plan and SOPs, the realistic path is 90 to 120 days. The first 30 days determine which timeline you are on.
What to Do in the First 30 Days After Getting an SQF Requirement from a Retailer
Most guides describe what SQF requires. Few explain what to do this week. Here is the sequence that works for small manufacturers without a dedicated food safety team.
Day 1-3: Confirm What Your Retailer Actually Requires
Before touching a document, make one call or send one email to your buyer contact. Ask:
- SQF Level 2 (GFSI-recognized) or will Fundamentals / FVA satisfy the requirement?
- Is there a retailer-specific addendum to the SQF audit?
- What is your deadline for certification before onboarding begins?
- Is there flexibility if certification is in progress but not yet complete?
This conversation can save weeks.
Day 3-7: Gap Analysis - Map What You Have Against What You Need
Most small manufacturers already have 40 to 60 percent of what SQF requires.

Do not try to fix anything in this phase. Map the gaps first. Clarity on what is missing is worth more than a half-finished System Manual.
Before you spend another week sorting folders, upload your HACCP plan, SOPs, supplier files, and production records into IONI. The system organizes what you already have, identifies missing SQF evidence, and turns scattered documents into a clear readiness map.
Day 7-10: Register in the SQFI Database and Appoint Your SQF Practitioner
Two steps. Neither can wait.
Register your site at sqfi.com. You need a site registration in the SQFI Assessment Database before you can schedule an audit with any certification body. SQFI registration fees range from $100 to $1,200 per year, depending on your annual sales.
Appoint your SQF Practitioner. This is a mandatory role - someone at your facility is accountable for developing, implementing, and maintaining the SQF system. The Practitioner must be employed by your site, hold a position of responsibility, and have completed a recognized HACCP training course. This can be an existing employee. It does not require a dedicated food safety hire.
Most small manufacturers lose 2 to 3 weeks because they treat these steps as something to do after the system is built. Do them now.
Day 10-20: Build Your Documentation System from Existing Materials
This is where most teams lose the most time. They open a blank SQF System Manual template and start writing their operation from scratch. Under a 90-day timeline, that approach runs out of runway.
The right starting point is what you already have. Your SOPs describe how your processes work. Your HACCP plan defines your hazards and controls. Your supplier documents contain most of what an approved supplier program needs. The gap is structure, not content.
Build in this order:
- HACCP plan: review against current process, add validation records, and annual review documentation
- SQF System Manual: the framework document that references all other procedures
- Supplier Approval Program: formalize existing supplier documents into an approved list with risk ratings
- Document control procedure: version numbering, review schedule, and access
- Internal audit schedule: at least one full internal audit planned before Stage 1

See how quickly your current documents can become an audit-ready SQF system. IONI connects HACCP, supplier approval, document control, monitoring records, and corrective actions in one workflow, so your team can start operating the system instead of building it manually.
Build my SQF system from existing documents →
Day 20-30: Start Collecting Records and Select Your Certification Body
By day 20, your system does not need to be finished. It needs to be running.
Start collecting records now. Every day your CCP monitoring logs are blank is a day you cannot recover. Day 20 is already late. Do not wait for perfect procedures before starting records. A system running with minor gaps and documented corrective actions is stronger than a perfect document set with two weeks of records.
Current outbreak investigations show why records matter. In a 2025 Salmonella Enteritidis outbreak linked to recalled eggs, FDA and CDC reported 134 illnesses across 10 states, 38 hospitalizations, and one death. For auditors and retailers, the lesson is practical: they want to see that monitoring, supplier controls, traceability, and corrective actions are working before a problem reaches the market.
Select your certification body (CB). A CB is the accredited organization that conducts your SQF audit. Contact two or three accredited CBs - get their pricing, current availability, and earliest scheduling windows. Based on multiple accredited CBs, a 2-day initial certification audit (desk + facility) typically runs $7,300 to $9,000 before travel expenses. Annual recertification audits run $4,500 to $6,000.
Do not schedule the audit yet. You need 90 days of records first. But knowing your CB's availability now tells you whether your timeline is realistic. A list of accredited CBs is available at sqfi.com under "Find a Certification Body."
Start collecting the records your auditor will ask for before your audit date is on the calendar. IONI helps your team capture CCP checks, supplier evidence, corrective actions, and audit records in a structured system from day one.
Start building audit records now →
How Long Does SQF Certification Take After a Retailer Requirement
The timeline depends on one thing: how much you already have.

The one variable you cannot compress is records. You cannot manufacture 90 days of consistent CCP monitoring logs in less than 90 days. Documentation you can accelerate. Records you cannot.
What happens if you miss a retailer's deadline?
It depends on the retailer. Some will grant a 30 to 60-day extension if you can show active progress: SQFI registration done, Practitioner appointed, CB selected. Others pause onboarding until the certificate is in hand.
The worst outcome is spending the first three weeks researching. That is how a 90-day timeline becomes a 150-day one. Contact your buyer proactively. Show your plan. Ask what is acceptable while certification is in progress.
Do You Need a Consultant to Get SQF Certified After a Retail Requirement?
No - under specific conditions.
You do not need a consultant if:
- You can appoint an internal SQF Practitioner
- You have an existing HACCP plan or the knowledge to build one
- You have SOPs that cover your main processes
- You are using a platform that structures documentation for you
You should consider a consultant if:
- You have no one internally who can serve as SQF Practitioner
- You have no HACCP plan and no experience building one
- Your timeline is under 60 days
- Your team has never worked with a food safety management system
Consultant fees typically run $2,500 to $12,000, depending on scope and the depth of support needed. That is a real cost, but not a required one. Most small manufacturers in the 10 to 100-employee range already have the knowledge internally. The bottleneck is structure and time, not expertise.
For teams replacing spreadsheets, modern systems significantly reduce dependency on consultants: Food Safety Software Overview
SQF Edition 10: What Changed and What to Prepare For in 2026
SQF Edition 10 was released in 2026, but SQFI states that audits to Edition 10 are anticipated to begin on January 2, 2027, and will not begin earlier than that date.
Until Edition 10 is recognized through the GFSI benchmarking process, Edition 9 remains the GFSI-recognized program for current audits. If your retailer deadline falls in 2027 or later, or if you are building a system you will maintain long-term, understanding Edition 10 now saves significant rework at your first recertification.
Key changes in SQF Edition 10 vs Edition 9:
- Weighted Core Clauses: Core Clauses now carry higher point deductions: confirm exact values with your certification body once Edition 10 is benchmarked. This makes Core Clause programs: food safety plan, GMP, CCP monitoring - significantly higher stakes.
- Food Safety Culture Assessment Plan: Sites must now have a formal, documented food safety culture plan with measurable objectives and evidence of improvement. Culture is no longer an informal expectation - it is auditable.
- Formal Change Management Clause: A documented process is now required to assess and control food safety risks from changes in people, processes, equipment, or suppliers.
- Environmental Monitoring: Now mandatory for all applicable sites and must be risk-based with documented justification.
- Streamlined structure: Restructured and streamlined for clearer navigation. Requirements were reorganized, not removed.
- Digital-first Code format: Edition 10 is delivered through a digital platform rather than a static document.
Score-based rating system: The letter ratings (Excellent, Good, etc.) are removed. Numerical scores remain for internal benchmarking and management review.

Practical implication for small manufacturers: If you certify in 2026 under Edition 9, plan a structured gap assessment against Edition 10 requirements by Q4 2026 - before your first recertification audit cycle begins.
Watch How IONI Helps with SQF Integration
FAQ
Does Whole Foods require SQF certification from all suppliers?
Whole Foods requires a food safety audit or certification from its suppliers. Most are directed toward GFSI-recognized certification, which means SQF Level 2.
Very small suppliers may qualify for the SQF Fundamentals Verification Assessment (FVA) - a remote verification that costs roughly 60% less than a standard on-site audit and takes approximately 60 days from scheduling to declaration. FVA is not a full GFSI certification and does not satisfy requirements where Level 2 is explicitly required. Confirm with your Whole Foods buyer contact before starting preparation.
What happens if I miss my retailer's SQF certification deadline?
It depends on the retailer and your contract terms. Some retailers grant a 30 to 60-day extension when suppliers can show active progress - SQFI registration complete, Practitioner appointed, certification body selected, with a scheduled audit date.
Others hold the onboarding start until the certificate is issued. Missing a deadline without communication almost always stalls the business relationship longer than the delay itself. Reach out proactively with a timeline before the deadline, not after.
Can I start selling to a retailer while my SQF certification is in progress?
Some retailers allow conditional onboarding while certification is in progress, particularly when you can demonstrate that your system is built and records are being collected. Walmart and Costco typically require a completed certificate before any supplier shipments begin.
Some regional retailers and Whole Foods have more flexibility for small suppliers during the certification period. Confirm directly with your buyer. Do not assume conditional approval is standard.
Is SQF certification required for all food products sold at Walmart?
Walmart requires GFSI-recognized certification for food and consumable suppliers. SQF Level 2 satisfies this requirement, as do BRCGS and FSSC 22000. The requirement applies to food product suppliers, not to non-food items.
Confirm the specific requirement for your product category in your Walmart supplier agreement or at walmart.com/supplier.
What is the difference between SQF Fundamentals and SQF Level 2 for retail suppliers?
SQF Fundamentals is an entry-level program covering basic GMPs and foundational food safety practices. It is not GFSI-recognized. Major retailers like Walmart, Costco, and Kroger require GFSI-recognized certification, which means Level 2 minimum. SQF Level 2 (Food Safety Code) is GFSI-benchmarked and is the standard most retailers require.
If your buyer says they need GFSI certification, they need Level 2. The only exception is Whole Foods, which may accept the Fundamentals Verification Assessment for qualifying small suppliers in specific categories.
How much does SQF certification cost for a first-time small manufacturer?
Total first-year costs typically include: SQFI site registration ($100 to $1,200 based on annual sales), certification body audit fees for a 2-day initial certification (desk + facility audit combined: $7,300 to $9,000 before travel), SQF Practitioner training, and internal preparation time.
Most small manufacturers spend $8,000 to $12,000 in direct out-of-pocket costs in the first year, excluding internal labor. Consulting fees, if used, add $2,500 to $12,000 depending on scope.
Can AI software help me get SQF certified faster after a retailer's requirement?
Yes. The main time cost in SQF preparation is documentation - building a HACCP plan, SQF System Manual, supplier approval program, and document control system. AI-powered platforms like IONI read your existing documents (SOPs, recipes, supplier specs) and build the SQF compliance structure automatically.
This compresses the documentation phase from several weeks to a few days, with your team reviewing and validating instead of writing from scratch. The 90-day records window is fixed - that cannot be shortened. But documentation, which typically takes 4 to 8 weeks manually, can be completed in days with the right tool.
The First 30 Days Set Your Entire Timeline
The retailers that require SQF do not move their onboarding windows. The 90-day records requirement does not shorten. Certification bodies cannot audit records that were never collected. What you can control is how quickly your team turns scattered documents, informal practices, and existing food safety knowledge into a working SQF system.
That is why the first 30 days matter so much.
By the end of month one, you should not be “researching SQF” anymore. You should have your retailer requirement confirmed, your SQF Practitioner appointed, your current documents mapped, your major gaps identified, your certification body options reviewed, and your daily records already running. Even if the system is not perfect yet, it should be operating.
For small manufacturers, this is usually the difference between a 90-to-120-day certification path and a six-month scramble. The companies that move fastest are not always the companies with the biggest QA teams. They are the ones that start with what they already have - HACCP plans, SOPs, supplier documents, COAs, production logs - and structure it into an auditable system before time is lost.
If a retailer just asked for SQF, the next 30 days matter more than any template you download. IONI turns your existing food safety documents into a working SQF readiness system, then helps your team collect the records auditors and buyers need to see.
Book a 30-minute SQF setup session →









