.png)
SQFI requires a minimum of 90 days of records before a site audit. Most manufacturers don't start collecting them until week eight.
That's the gap. Not knowledge, not intention — timing. A buyer from Whole Foods, Costco, or Walmart approves your product and adds one condition: SQF certification before onboarding. You now have a fixed window and a team that's still running production.

Most guides explain what SQF requires. This one tells you what to do this week, what can wait, and what breaks the timeline if you get the sequence wrong.
What SQF Certification Actually Requires (And What Auditors Check First)
SQF certification is often misunderstood as a documentation exercise. In reality, it is a system validation process. Auditors are not evaluating whether you have policies written down. They are evaluating whether your operation consistently produces safe, controlled, and traceable outputs.
When an SQF audit starts, the first interactions usually determine the outcome. Auditors do not begin with general discussions. They go directly into your system. They request your HACCP plan, then move into monitoring records, corrective actions, supplier approvals, and traceability documentation. Within the first hour, they can tell whether your system is real or theoretical.
What they are looking for is alignment between three layers. The first is structure, which includes your HACCP plan and SQF System Manual. The second is execution, which is reflected in your day-to-day records. The third is response, meaning how your system handles deviations through corrective actions and verification.
Small manufacturers rarely fail because they lack knowledge. They fail because these three layers are disconnected. Records might exist, but they are inconsistent or filled retrospectively. Corrective actions might be logged, but they do not include root cause or verification.
This gap between documentation and reality is what auditors identify quickly. It is also why systems built on spreadsheets or disconnected tools tend to break under audit conditions.
If you look at how SQF fits into broader certification frameworks, it becomes clear why this matters. SQF is part of the GFSI ecosystem, which emphasizes not just compliance, but operational proof. If you want a deeper understanding of how SQF compares to other schemes, you can explore What are GFSI schemes and how to choose the right one.
Is 90 Days Realistic? The Honest Answer
A 90-day SQF certification timeline is realistic, but only under specific conditions. The biggest misconception is that documentation is the main bottleneck. In practice, documentation can be created relatively quickly, especially if you already have SOPs and operational procedures in place. The real constraint is the time required to generate consistent, audit-ready records.
Auditors expect to see evidence over time. They want to review logs that show how your system behaves under normal conditions, not just during audit preparation. This includes CCP monitoring records, sanitation logs, supplier validations, and corrective actions. If you start collecting these records too late, you simply run out of time.

Most teams follow a sequence that makes 90 days impossible. They spend the first six to eight weeks writing documentation, often from scratch, and only begin implementing the system afterward. By the time they reach internal audit, they realize they only have one or two weeks of usable records.
That is not enough to demonstrate consistency.
Your 90-Day SQF Preparation Plan, Week By Week
To make 90 days work, the sequence has to change. Documentation needs to be built quickly, using existing materials wherever possible. Record collection needs to start early, even if the system is not perfect yet. The focus should be on creating a functioning system as soon as possible, then refining it, rather than trying to perfect documentation before implementation.
Weeks 1-2: Gap Analysis and Documentation Audit
The first two weeks set the direction for everything that follows. The most common mistake at this stage is starting from a blank page. In reality, most facilities already have a large portion of the required information. It is just scattered across different formats, locations, and levels of completeness.
During these weeks, the focus is on collecting and mapping, not writing. You start by gathering all existing materials, including SOPs, production logs, cleaning procedures, supplier documents, and training records. This often involves pulling files from shared drives, email attachments, and even printed documents.
Once everything is collected, the next step is mapping it against SQF requirements. This is where you build your working SQF certification checklist. For each clause in the SQF Code, you identify whether you have full coverage, partial coverage, or no coverage at all. This exercise quickly highlights gaps, but it also reveals how much you already have.
At the same time, you define your process flow. This includes every step from receiving raw materials to final product distribution. This process flow becomes the backbone of your HACCP plan. Without it, hazard analysis cannot be structured correctly.

You also need to assign your SQF Practitioner during this phase. This is a required role and cannot be treated as a side responsibility. The practitioner is accountable for maintaining the system, coordinating with auditors, and ensuring implementation across the facility.
Many teams lose time in these two weeks by trying to perfect documents instead of identifying gaps. A more effective approach is to move quickly through mapping and use tools like IONI to structure existing documentation automatically.
By the end of week two, you should not have a finished system. You should have clarity on what is missing and what needs to be built.
Weeks 3-6: Build Your HACCP Plan And SQF System Manual
This phase is where most of the technical work happens. Your HACCP plan defines how your facility identifies and controls hazards. It is the core of your SQF system, and everything else connects to it.
Building a HACCP plan involves analyzing each process step, identifying potential hazards, assessing risk levels, defining control measures, and determining critical control points. For each CCP, you also need to define monitoring procedures, corrective actions, and verification steps.
For example, in a cooking process, the primary hazard might be pathogen survival. The control measure is achieving a specific minimum temperature. The CCP is the cooking step itself. Monitoring involves measuring temperature for each batch, and corrective action might involve reprocessing or discarding the product if the target is not met.
This level of detail needs to be applied across your entire process flow. It is not a difficult concept, but it is time-consuming if done manually.
Most facilities already have the underlying knowledge. Operators know where risks occur. SOPs describe how processes are controlled. The challenge is structuring this information into a compliant HACCP format.
This is where modern systems significantly reduce effort. Platforms like IONI, developed for food manufacturers, allow you to upload existing documents and generate structured HACCP plans automatically. Instead of writing everything from scratch, your team reviews and validates the output.
At the same time, you build your SQF System Manual. This includes sections on document control, traceability, corrective actions, supplier management, and management responsibility. Many of these sections overlap with your HACCP plan and can be developed in parallel.

Teams that rely entirely on manual processes often spend four to six weeks in this phase. Teams that reuse existing documentation and structure it efficiently can complete it much faster.
Already have SOPs or an existing HACCP plan? Upload your documents and see your SQF system built in one session — no consultant, no manual entry.
Weeks 7-9: Implement and Start Collecting Records
Once your system is defined, the focus shifts to implementation. This is where theoretical compliance becomes operational reality. It is also where most timelines fail.
Auditors expect to see consistent records across multiple weeks. These records include CCP monitoring logs, sanitation checks, supplier validations, deviation reports, and corrective actions. It is not enough to have forms filled out. The records must show that your system is being followed consistently across shifts and over time.
One of the most important aspects of this phase is starting early. Even if your documentation is not perfect, you need to begin collecting records as soon as possible. Waiting until everything is finalized reduces your available data window and weakens your audit position.
Operationally, this phase introduces new challenges. Operators need to follow updated procedures, log data correctly, and respond to deviations. Supervisors need to review records regularly, not just before audits. QA teams need to ensure that corrective actions are documented and verified.

Supplier management also becomes critical at this stage. Certificates of Analysis need to be reviewed against specifications, and supplier approvals need to be current. This is one of the most time-consuming manual tasks in many facilities.
Using Ingredients Intelligence for food companies helps automate COA validation and supplier checks, reducing the burden on QA teams and improving consistency.
The goal of this phase is not perfection. It is consistency. Your system needs to show that it works under normal operating conditions.
Weeks 10-11: Internal Audit and Corrective Actions
At this point, you have a working system and several weeks of records. The next step is to test it under audit conditions.
A proper internal audit is not a checklist exercise. It is a simulation of the external audit. You select a batch and trace it through the entire process, from raw materials to final product. You verify supplier documentation, review production logs, check CCP monitoring, and confirm traceability.
This process usually reveals issues. Records might be missing for certain days. Logging might be inconsistent between shifts. Corrective actions might be documented but not verified. Supplier documents might be outdated.
These findings are not a failure. They are expected. What matters is how you respond to them.

Each issue needs to go through a full corrective action cycle. This includes identifying the root cause, implementing a corrective action, and verifying that the issue has been resolved. For example, if a CCP deviation was not recorded, the root cause might be insufficient operator training. The corrective action could involve retraining and updating procedures, and verification might include supervisory review over the following weeks.
Auditors expect to see this cycle. A system with no recorded issues often raises more concern than a system with documented and resolved issues.
Week 12: Desk Audit Prep & Final Readiness Check
The final week focuses on preparing for the desk audit. This is the stage where auditors review your documentation remotely and decide whether your facility is ready for the site audit.
You need to submit your HACCP plan, SQF System Manual, key procedures, and sample records. The emphasis is on completeness and consistency. Documents need to align with each other, and records need to support the procedures described.
This is also the point where gaps become visible if they were not addressed earlier. Missing records, inconsistent logs, or incomplete corrective actions can delay the process.

Modern systems that continuously validate documentation and records can significantly reduce risk at this stage. For example, approaches described in The role of AI in modern SFCR compliance show how real-time validation helps maintain audit readiness.
By the end of week twelve, your goal is not perfection. It is confidence that your system is consistent, traceable, and defensible.
The 4 Documents Auditors Pull First. And Where Small Manufacturers Fail
Across SQF audits, there is very little randomness. Most auditors follow the same path because it gives them the fastest signal on whether your system actually works. The FDA estimates that inadequate monitoring and recordkeeping are among the most common causes of non-compliance in food safety systems, frequently cited in inspection observations.
They start with four things: your HACCP plan, CCP monitoring records, corrective actions (CAPA), and supplier documentation.
This is not a checklist. It is a diagnostic. Within 30- 60 minutes, these four areas tell the auditor whether your system is real or just documented.
The HACCP plan is the starting point because it defines how your operation is supposed to work. The auditor is not reading it for theory. They are checking whether it reflects your actual process flow, real hazards, and real control points. From there, auditors move to CCP monitoring logs. This is where most systems start to break. They are looking for consistency across time and across people. Do different operators log data the same way? Are there gaps in records? Do the numbers look realistic, or are they always “perfect”? A week of flawless logs often raises more concern than a week with a documented deviation and a clear response.
Next comes corrective actions. This is where the system either proves itself or fails. Auditors expect to see that when something goes wrong, it is identified, investigated, corrected, and verified. What they often find instead are superficial entries: “issue corrected” with no explanation of why it happened or how recurrence is prevented. Without root cause and verification, CAPA is incomplete, even if the form is filled out.
Finally, supplier documentation. This is usually where external risk shows up. Auditors check whether suppliers are approved, whether Certificates of Analysis are current, and whether incoming materials are verified against specifications. The common failure here is not missing documents, but outdated or unreviewed ones. Files exist, but no one can show when they were last checked or how they are validated against requirements.
The pattern across all four areas is the same. The issue is not missing information. It is a lack of connection. The HACCP plan does not match the logs. The logs do not trigger meaningful corrective actions. Supplier controls are not tied back to specifications.
That is why systems built on disconnected documents struggle under audit conditions. The Food Safety platforms, like IONI or FoodReady, may handle this. However, the difference is not in features or templates. It is whether your documentation, records, and decisions are linked into one system that reflects how your operation actually runs.
Watch How IONI Works:
This is exactly the gap IONI closes. Upload your documents and see your audit-ready system in 30 minutes. No credit card required. Setup included.
How To Build A Plan Without Spending Weeks Starting From Scratch (IONI)
Most teams lose the first two weeks staring at a blank template. It's the wrong starting point.
You already have most of what you need: SOPs, production logs, supplier specs, and existing procedures. The problem isn't missing information. It's that everything lives in separate places: shared drives, email, printed binders.

IONI changes the starting point. You upload the documents you already use. The platform reads them and builds your compliance system: HACCP plan, operator checklists, supplier tracker, structured for SQF, BRCGS, or FSMA. Your team reviews and refines instead of drafting from zero.
What usually takes weeks of writing becomes a few days of validation. More importantly, the system reflects how work is actually done on your floor, so when auditors compare procedures to records, they see alignment, not gaps.
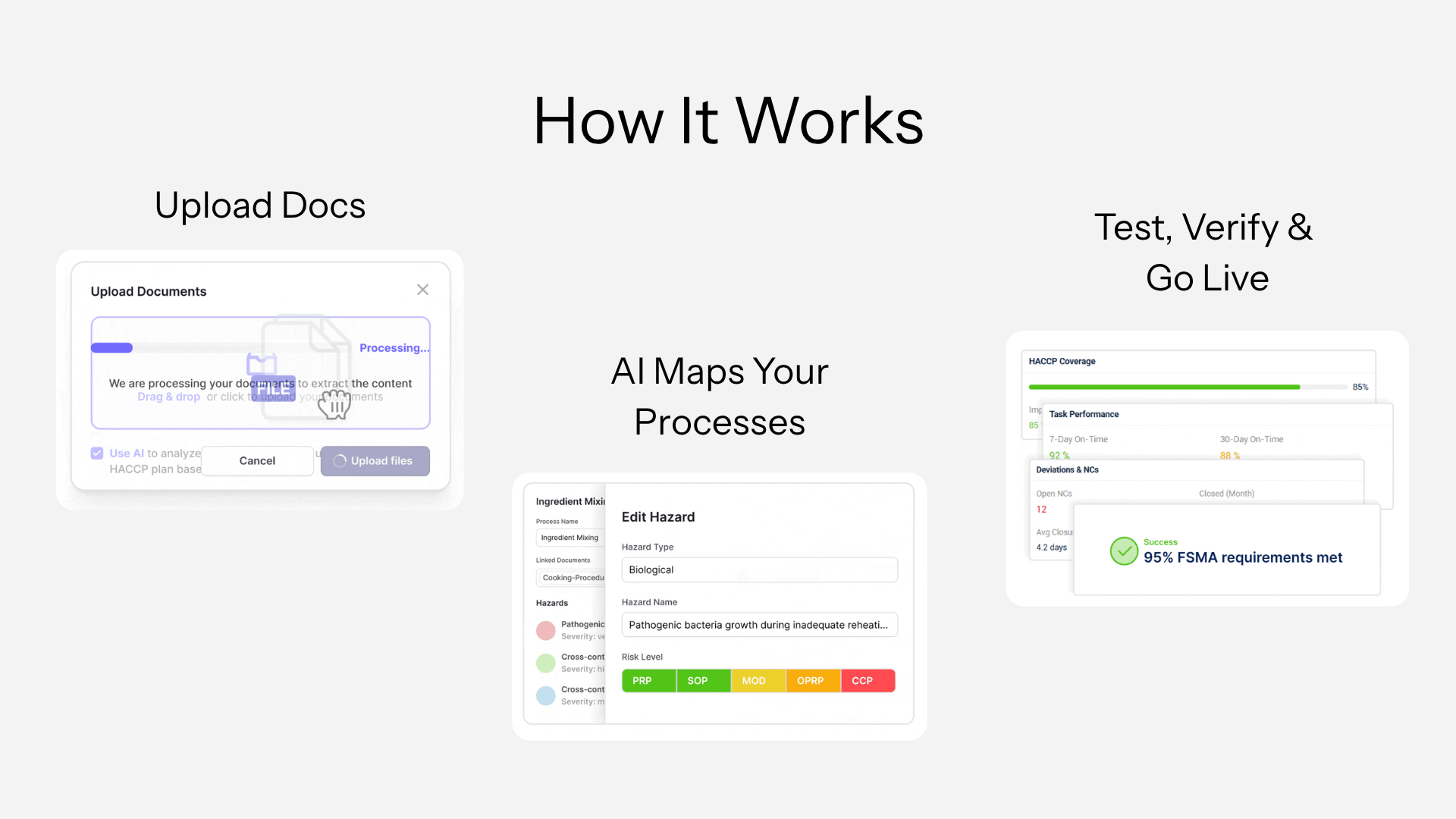
Monitoring works the same way. Operators log checks directly in the system. Each entry is validated in real time against defined limits. If something goes outside tolerance, a corrective action starts immediately, not after someone notices a problem at end of day.

Supplier control is handled through Ingredients intelligence: COA mismatches are flagged automatically before materials move into production. Regulatory changes are tracked through AI regulatory intelligence and translated into updates before your next audit.
Everything connects in one place, as outlined in Food manufacturing software solutions. Not separate pieces. One workflow that reflects how your operation runs.
See how this works with your own documents.
What To Do If You’re Already Behind Schedule
If you’re behind, the instinct is to push harder and try to fix everything at once. That usually makes it worse. The faster path is to narrow focus and stabilize what actually matters for the audit.
Start by accepting that you won’t perfect the entire system before certification. You don’t need to. You need a system that works consistently within a defined scope.
The priority order should be clear and non-negotiable. First, make sure your core plan is complete and reflects your real process. Without that, everything else becomes disconnected. Second, start collecting records immediately, even if parts of the system are still being refined. Time lost here cannot be recovered later. Third, focus on the areas auditors rely on most: monitoring at critical points and how you handle deviations. If these are weak, the rest of the system won’t compensate.
If pressure is high, reduce complexity instead of adding effort. That often means narrowing the scope of certification. Fewer product lines, fewer process variations, tighter boundaries. A smaller, stable system passes audits more reliably than a broad system that is only partially implemented.
This is also where modern tools can change the trajectory. Instead of trying to manually close every gap, systems that structure your documentation, validate records in real time, and highlight missing elements help you recover lost time quickly. The broader shift toward this kind of approach is outlined in How AI is transforming food manufacturing in 2026, where compliance moves from reactive work to continuous validation.
What auditors are trying to determine is simple. Does this system work in practice, and does the team follow it consistently? They are not scoring how much you built. They are assessing whether what you built holds up under normal conditions.
A focused, consistent system will pass. A wide, incomplete one won’t.
FAQ
How Long Does SQF Certification Take For A First-Time Applicant?
Most first-time SQF certification timelines fall between three and six months. The difference comes down to how quickly you move from documentation to real execution. A 90-day timeline is achievable when documentation is built quickly, and record collection starts early. If records only begin near the end, timelines almost always extend.
Can A Small Manufacturer Get SQF Certified Without Hiring A Consultant?
Yes, and it’s more common than it used to be. Small teams often handle SQF audit preparation internally by using structured frameworks and software that guide the process. The key requirement is having a capable SQF Practitioner and a system that keeps documentation and records consistent. Consultants can accelerate progress, but they are not required for certification.
What Records Does An SQF Auditor Want To See Before The Site Audit?
Before the site audit, auditors focus on whether your system produces consistent evidence. That includes your HACCP documentation, CCP monitoring logs, corrective actions, supplier approvals, and traceability records. They are not just checking that records exist. They are checking whether they are complete, consistent over time, and aligned with your procedures.
What Is The Most Common Reason Small Food Manufacturers Fail Their First SQF Audit?
The most common failure point is not missing documentation. It is inconsistent. Records are incomplete, corrective actions are not fully documented, or procedures do not match what is happening on the floor. Auditors look for a system that works in practice, not one that looks correct on paper.
How Much Does SQF Certification Cost For A Small Manufacturer?
Costs vary depending on the certification body, facility size, and how preparation is handled internally. Typical costs include audit fees, certification fees, and the internal time required to build and run the system. Additional costs may come from consulting or software support.
Do I Need A Separate HACCP Certification Before Getting SQF Certified?
No. HACCP is built into the SQF framework. You do not need a separate certification, but your HACCP-based system must meet SQF requirements and be fully implemented and documented.
Can I Use AI To Build My System For SQF Certification?
Yes. AI tools can structure your existing documentation, identify gaps, and help you build a compliant system faster. They are especially useful for reducing manual work and accelerating early phases. However, responsibility still sits with your SQF Practitioner, who must review, validate, and ensure the system reflects actual operations.
If your audit is in the next 90 days, the documentation phase is where most timelines break. Start there.



.png)
.png)




