
Introduction
Compliance teams tracking regulatory changes manually run into the same problem regardless of industry: updates arrive after they matter. A guidance document revises a CCP limit. A labeling requirement changes for a key export market. An FDA draft guidance signals where enforcement is heading. By the time the information reaches the team through newsletters, consultant briefings, or manual website checks, the window for proactive response has often already closed.
AI regulatory intelligence tools change the dynamic. They monitor regulatory sources continuously, filter for relevance to your specific operation, and surface what requires action. This guide compares the leading platforms in 2026 on the dimensions that determine real-world usefulness: source coverage, analysis depth, document integration, and cost.
In this article, we highlight the leading AI regulatory intelligence tools in 2026 and how they help businesses maintain compliance, reduce risk, and optimize regulatory workflows.
What AI Regulatory Intelligence Software Does
Regulatory intelligence software automates three things compliance teams currently do manually:
1. Monitoring
Instead of manually checking regulatory websites on a rotating basis, your team receives alerts when new rules, amendments, or guidance are published. Coverage extends beyond primary regulators to include national food safety authorities, regional agencies, and international standard bodies that are rarely monitored consistently in manual workflows.
2. Analysis
New regulatory updates are classified, prioritized, and assessed for relevance to your operation. AI systems map changes against your internal documentation, including SOPs, policies, and process descriptions, and identify exactly what requires review or revision.
3. Action
Leading platforms connect regulatory updates directly to internal workflows. Documents are flagged for update, draft revisions are generated, and tasks are assigned to responsible team members. The time between identifying a regulatory change and acting on it is reduced from days to hours.
Market Context
The Regulatory Technology market is projected to grow from approximately 19.6 billion dollars to over 82 billion dollars by 2032, reflecting a compound annual growth rate of about 22.8 percent. Industry surveys show that more than half of companies have adopted basic AI driven compliance tools, while the most mature organizations increasingly rely on automated systems for continuous regulatory monitoring and response.
Quick Comparison: 6 AI Regulatory Intelligence Platforms
Choosing the right regulatory compliance platform depends on how your team works and how much automation you need. Some tools focus on monitoring specific sources, while others provide full compliance systems with AI-driven analysis and document mapping.
The comparison below highlights how leading platforms differ in scope, target users, and pricing, helping food, pharma, and compliance teams evaluate the right fit.
.png)
Not all regulatory tools solve the same problem.
Lightweight tools focus on tracking changes, while enterprise solutions combine monitoring with consulting and manual workflows. AI-first platforms like IONI stand out by connecting regulatory intelligence directly to internal documents and compliance processes.
For teams that need more than alerts, the key difference is moving from passive monitoring to active compliance management with automated analysis, faster onboarding, and scalable operations.
#1 IONI: AI Regulatory Intelligence for Food, Pharma, and Compliance Teams
IONI monitors regulatory sources across jurisdictions: EU Lex, FDA, CFIA, industry-specific frameworks, and converts updates into structured data mapped to your internal documents. The key differentiator from monitoring-only tools: IONI doesn't just tell you something changed. It tells you which of your SOPs, policies, and processes are affected, and flags what needs updating.
Source configuration is tailored per account: operating regions, applicable standards, relevant frameworks. When a new or updated regulation comes in, an AI agent assesses applicability, adds relevant documents to a private regulatory library, and breaks requirements into discrete elements mapped to internal processes. Compliance teams see what changed and what it means for their specific operation.
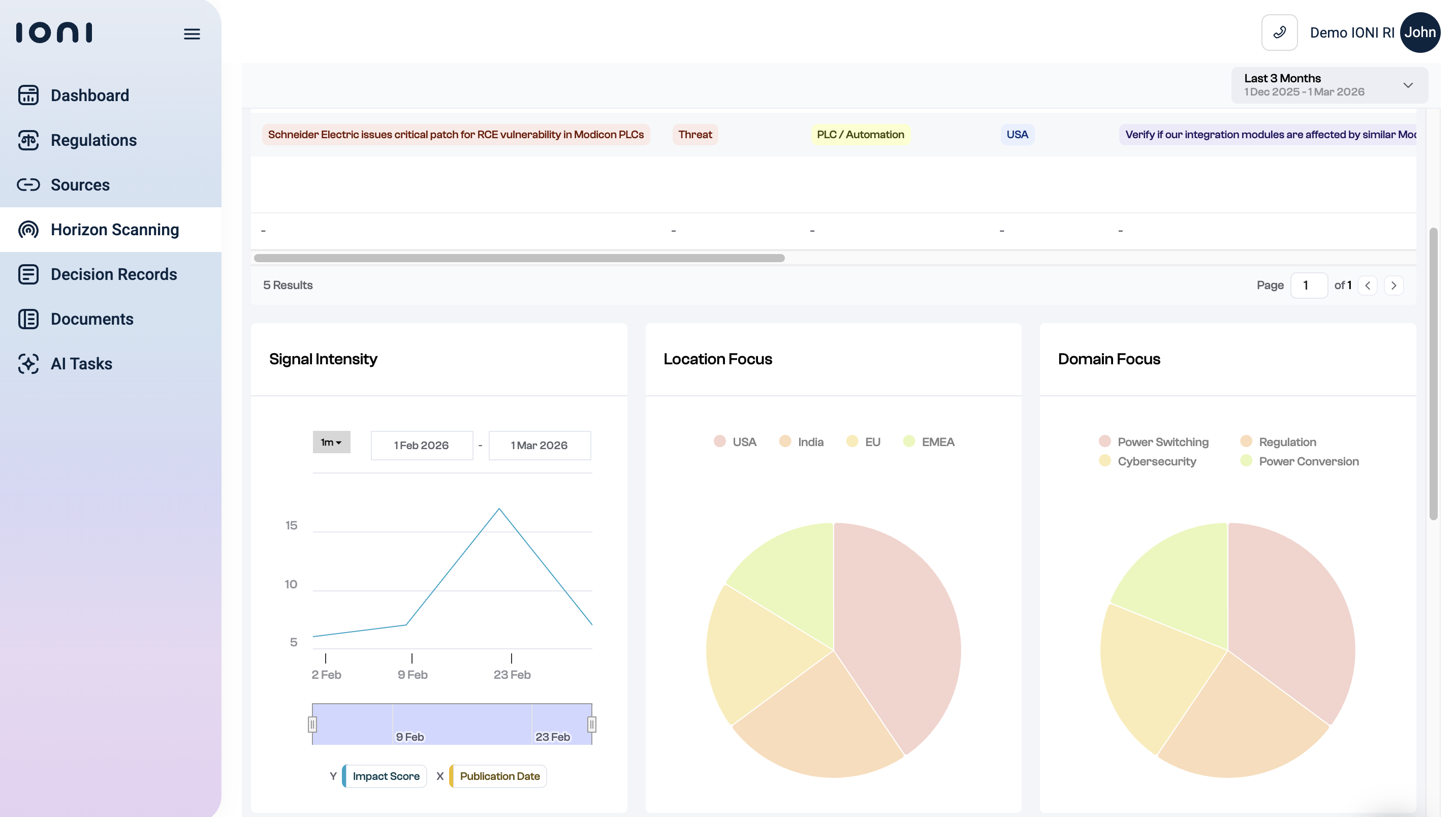
Key Features
- Live compliance monitoring: Regulatory changes tracked across jurisdictions and standards including Codex, FDA, EU, CFIA, GFSI, and ISO.
- AI regulatory search: Complex compliance questions answered with cited responses against your specific regulatory library.
- Change alerts with impact analysis: Notifications that include which internal documents are affected, not just that a change occurred.
- Gap and risk detection: Compliance gaps flagged before they appear in audits.
- Document auto-mapping: Regulatory requirements linked directly to contracts, policies, and SOPs.
Who it's For
- Food and beverage manufacturers managing HACCP compliance, labeling standards, and multi-market food safety requirements.
- Life sciences and pharma companies managing evolving health and safety regulations across jurisdictions.
- Compliance teams responsible for multi-jurisdictional tracking across legal, quality, and operations functions.
Watch How IONI Works
Pricing
- Food Safety module (includes HACCP Builder): from $199/month
- Regulatory Intelligence module: contact for pricing
- Enterprise: custom
- GDPR-compliant; SOC 2 certification - in progress.
See how IONI maps regulatory changes to your internal documents. Book a demo now!
#2 Visualping: Web Change Monitoring for Regulatory Teams
Visualping is not a regulatory intelligence platform. It is a page monitoring tool used by compliance teams to track changes on specific websites such as government portals, regulatory agency pages, and official publication sources.
This distinction is important. Visualping does not interpret regulatory content, assess its impact on your operation, or connect changes to your internal documentation. It detects that a page has changed, highlights the difference, and provides a plain language summary. The analysis and follow up remain the responsibility of your team.
For teams that need to monitor a defined set of sources, Visualping is effective. This includes pages such as a national food safety authority guidance section, an EU regulatory portal, or a specific FDA docket. It can be deployed quickly, requires minimal setup, and operates reliably.
The tool monitors any URL at selected intervals and sends alerts when changes occur, typically including a visual comparison with highlighted updates. It is also significantly more cost efficient than full regulatory intelligence platforms, making it a practical option for targeted monitoring use cases.
Key Features
- Monitors any web page, including government portals, PDFs, and social media
- AI-generated plain-English summaries of what changed, with an IMPORTANT flag for relevance
- Configurable check frequency: every 5 minutes to once weekly
- Four detection modes: visual, text, element, and combined (All mode recommended)
- Business plan integrations: Slack, Teams, Google Sheets, API
Who it's For
Teams needing lightweight monitoring of specific regulatory pages without the overhead of a full compliance platform. Works well alongside a dedicated regulatory intelligence tool, not as a replacement.
Pricing
- Free plan: 5 pages, 150 checks/month, 60-minute minimum interval. AI summaries included on all plans.
- Personal plans: from $10/month (1,000 checks, 25 pages) to $50/month (10,000 checks, 200 pages). Email alerts only.
- Business plans: from $100/month (20,000 checks, 500 pages) to $250/month (50,000 checks, 1,500 pages). Includes Slack, Teams, Google Sheets integration.
#3 DDReg Pharma RegIntel: Regulatory Intelligence for Life Sciences
DDReg Pharma's RegIntel is built for pharmaceutical and life sciences companies managing complex global regulatory requirements. It provides a cloud-hosted portal with real-time tracking of regulatory updates, document comparison across markets, and lifecycle management for post-approval variations.
Unlike general-purpose monitoring tools, RegIntel is designed around pharma-specific workflows: tracking health authority guidance, managing product lifecycle compliance, and providing strategic advisory support for market entry. It's not positioned for food manufacturers or general compliance teams.
Key Features
- Real-time tracking: Global regulatory updates monitored continuously.
- Instant notifications: Alerts on changes relevant to specific operations.
- Document comparison: Cross-regional comparison to identify compliance inconsistencies.
- Lifecycle management: Post-approval variation tracking and ongoing compliance monitoring.
- Strategic advisory: Expert support for regulatory strategy development.
Who it's For
Pharmaceutical companies, biotech firms, medical device manufacturers, and regulatory affairs professionals.
Pricing: By quote. Contact DDReg directly.
#4 Freyr RegIntel: Regulatory Intelligence for Life Sciences and Food
Freyr RegIntel covers a broader industry range than most platforms in this list: pharmaceuticals, medical devices, cosmetics, food and dietary supplements, and biocides. Its AI assistant, Freya.Intelligence, answers regulatory questions against a database of over 95,000 verified regulations from 200+ markets.
The platform suits teams that need to monitor regulatory changes across multiple product categories and markets simultaneously, with query-based access to regulatory data rather than just alert feeds.
Key Features
- Freya.Regulations: 95,000+ verified regulations from 200+ markets, updated continuously by 2,300+ web-crawling bots and expert validation.
- Freya.Alerts: Customizable regulatory alerts with direct links to source documents.
- Freya.Dashboards: AI-driven dashboards for trend analysis and compliance gap identification.
- Freya.RTQ: Regulatory query handling with AI-powered response drafting from emails, PDFs, and images.
Who it's For
Pharmaceutical companies, medical device manufacturers, cosmetics and food supplement companies, biocide producers. Strong fit for multi-market regulatory affairs teams.
Pricing
- Free trial: 14 days
- Plus Plan: from $100/user/month
- Corporate: custom pricing
#5 Deloitte RegAI: Enterprise Regulatory Intelligence
Deloitte's RegAI is a generative AI platform for large organizations that need to interpret regulatory requirements, assess gaps against existing policies, and generate compliant procedures at scale. It's designed for multinationals with complex, multi-jurisdictional compliance obligations — not for SMEs or single-market operators.
Unlike purpose-built regulatory intelligence tools, RegAI sits within Deloitte's broader consulting ecosystem. That means strong professional services support alongside the software, and enterprise pricing to match.
Key Features
- Regulatory interpretation: AI-assisted understanding of requirements, gap assessment against existing policies, and draft policy generation.
- Automated gap assessment: Identifies discrepancies between current policies and new regulatory requirements.
- Policy generation: Drafts and updates policies to align with evolving standards.
- Multilingual support: All major languages, including all 24 official EU languages.
Who it's For
Financial institutions, life sciences organizations, and large multinational corporations. Not suitable for food manufacturers, SMEs, or teams seeking self-service regulatory monitoring.
Pricing: Enterprise consulting engagement. Contact Deloitte directly.
#6 RegASK: Regulatory Intelligence with Expert Network Access
RegASK combines AI-driven regulatory monitoring with direct access to over 1,700 regulatory subject matter experts across 120+ countries. For regulations where AI-generated analysis isn't sufficient — complex market access questions, technical standards in emerging markets, niche product categories — human expert access is a real differentiator.
The platform covers life sciences, food and beverages, personal care, animal care, and household chemicals. RegAlerts delivers curated regulatory updates globally; RegInsights provides analytics and trend analysis; RegGenius is the conversational AI interface for regulatory questions.
Key Features
- RegGenius: Conversational AI interface for regulatory questions with AI-driven translation.
- RegAlerts: Customized global regulatory monitoring with task assignment and workflow support.
- RegInsights: Analytics, horizon scanning, and interactive regulatory mapping across markets.
- AskRegASK: Direct access to 1,700+ regulatory experts in 120+ countries for complex queries.
Who it's For
Life sciences, pharma, biotech, medical devices, food and beverages, personal care, and animal care companies — particularly those in emerging markets or dealing with regulations that require expert interpretation.
Pricing: By quote. Expert consultation fees may apply separately.
To sum up. Most regulatory tools fall into three categories.
Monitoring tools like Visualping are fast to set up but limited to tracking changes without deeper analysis. Industry-specific platforms like Freyr and DDReg provide broad regulatory coverage, but are often complex, expensive, and rely on manual workflows. Enterprise solutions like Deloitte RegAI add consulting and customization, but come with longer timelines and higher costs.
AI-first platforms like IONI take a different approach by connecting regulatory intelligence directly to your internal documents and processes. Instead of just tracking changes, they analyze impact, map requirements, and support execution, making them better suited for teams that need operational compliance, not just visibility.
See how IONI monitors regulatory changes and maps them to your internal documents automatically.
What To Look For When Evaluating Regulatory Intelligence Software
Coverage, automation, and alert delivery are baseline capabilities. Most platforms at this level offer them. The differences that drive long term value are less visible and often only become clear after implementation.
Source flexibility. Many tools restrict you to a fixed set of regulatory sources. This works for broad coverage but creates gaps in niche markets or less standardized regions. Check whether the platform allows custom sources. Some systems support configuration at the account level, while simpler tools monitor any URL but do not structure the data. The ability to define and adapt sources is critical if your regulatory scope is not static.
Relevance scorin. Without prioritization, alerts become noise. A platform that flags every update across all sources increases workload instead of reducing it. Look for systems that evaluate relevance based on your products, processes, and operating regions. Effective scoring reduces volume and focuses attention on changes that require action.
Audit trail. A complete audit trail is essential. The platform should record who reviewed each update, when it was assessed, and what action was taken. This is not only useful for internal tracking but also necessary to demonstrate due diligence during audits or regulatory reviews.
Document mapping. Regulatory updates need to be connected to the documents that govern your operations. Platforms that map changes directly to SOPs, policies, and procedures remove a significant amount of manual work. Without this, teams still need to interpret each change and identify affected documents themselves.
Historical trend analysis. Basic tools show what changed today. More advanced platforms provide visibility into patterns over time. This includes tracking which regulators are most active, which topics are evolving fastest, and where future risk is likely to emerge. This shifts compliance from reactive updates to structured planning.
AI explainability. Recommendations must be clear and defensible. If a system suggests an action without explaining the reasoning, it introduces risk. Teams need to understand why a change matters before they can implement it, and they must be able to justify decisions during audits.
Data ownership. Before selecting a platform, confirm how your data is handled. You should be able to export your regulatory library, assessments, and audit logs in standard formats such as CSV or PDF, or access them via API. It is equally important to understand what happens to your data if you decide to switch providers.
In practice, the evaluation should focus less on feature lists and more on how the platform supports real compliance workflows over time.
Which Platform Fits Your Situation
Different compliance tools are built for different levels of complexity. Some are designed for simple monitoring, while others support full regulatory intelligence, document mapping, and enterprise-scale compliance workflows.
The table below helps you quickly match your specific needs with the platform that best fits your use case.

The right choice depends on how deep you need to go.
If you need simple tracking, lightweight tools are enough. If you operate in regulated industries like food or pharma and need to connect regulations to internal documents and processes, AI-driven platforms provide significantly more value.
At a higher level, enterprise and expert-driven solutions add scale and support, but with increased cost and complexity. The key distinction is whether you need visibility, guidance, or full execution of compliance.
Conclusion
KeepinRegulatory intelligence software spans a wide range, from simple page monitoring tools to fully integrated AI driven compliance systems. The right choice depends on the specific problem your team needs to solve.
Visualping is suited for teams that need straightforward monitoring of specific regulatory pages with minimal setup. It tracks changes and alerts your team, but does not interpret or operationalize them.
DDReg, Freyr, and RegASK are designed primarily for life sciences and pharmaceutical compliance. Their strengths lie in curated regulatory databases, access to domain experts, and structured analytics across global markets.
Deloitte RegAI targets large enterprises with complex, multi jurisdictional compliance requirements. It offers deep integration and advisory capabilities, typically aligned with organizations that have dedicated compliance teams and significant budgets.
For food manufacturers operating under frameworks such as FSMA, CFIA, SQF, BRCGS, and FSSC 22000, IONI combines regulatory monitoring with document level mapping, gap detection, and audit ready outputs within a single system.
See how IONI connects regulatory changes to your documents. Book a demo.
FAQ
Can Regulatory Intelligence Tools Handle Updates From Multiple Countries Simultaneously?
Yes. Multi jurisdiction monitoring is a core capability of modern platforms. IONI, Freyr RegIntel, and RegASK each support coverage across 200 or more markets. Deloitte RegAI also supports all official EU languages.
The real difference is not coverage, but how effectively each platform filters and prioritizes updates based on your products, regions, and regulatory scope.
Are These Tools Suitable For Small Or Mid Sized Businesses Or Only Large Enterprises?
It depends on the platform. Visualping offers a free entry point. Freyr RegIntel starts at around 100 dollars per user per month. IONI starts at 99 dollars per month for food safety use cases.
Enterprise solutions such as Deloitte RegAI are typically designed for large organizations with more complex requirements and budgets. For small and mid sized food manufacturers, IONI, Freyr, or Visualping are the most practical starting points.
How Do Platforms Ensure Accuracy And Reliability Of Regulatory Data?
Reliable platforms source data directly from official regulators such as EUR-Lex, FDA, and national food safety authorities. Updates are processed through AI systems and, in some cases, validated by human experts.
IONI includes manual review as part of the workflow. Freyr combines expert validation with automated monitoring at scale. For critical decisions, verification against the original regulatory source remains best practice.
Can IONI Integrate With Existing Systems And Workflows?
Yes. IONI supports integration via API and CSV and is designed to work alongside existing tools. Its AI modules connect regulatory changes to internal documents and generate actionable outputs within your current compliance processes.
What Is The Difference Between Regulatory Intelligence Software And Compliance Management Software?
Regulatory intelligence focuses on monitoring external changes, assessing relevance, and informing teams about what requires action. Compliance management focuses on execution, including documentation, corrective actions, and audit readiness.
IONI combines both functions in one system, while most platforms in this category focus primarily on regulatory monitoring.
How Quickly Can A Team Get Started With AI Regulatory Intelligence Software?
Setup time varies by platform. Visualping can be configured in minutes by adding a URL and selecting a monitoring frequency. IONI typically requires initial configuration of regulatory sources and document upload, with most teams operational within a few days.
Enterprise platforms such as Deloitte RegAI involve longer implementation cycles, often measured in weeks, in exchange for deeper integration and customization.
Related Reading
Best HACCP Software for Food Manufacturers in 2026
How to Create a HACCP Plan: Step-by-Step Guide for Food Manufacturers
Manual vs. AI-Generated HACCP Plans: What Actually Saves Time and Passes Audits
The Future of Food Safety: How AI Is Changing HACCP Compliance









