.png)
Introduction: Why Supplier Quality Is The #1 Food Safety Risk And Why This List Is Different
Supplier quality failures remain the most consistent root cause behind recalls, audit non-conformances, and rejected shipments in food manufacturing.
The financial exposure is not theoretical. Industry data shows that the average food recall costs approximately $10 million in direct expenses alone, with total impact often exceeding $30–50 million when factoring in brand damage, lost contracts, and operational disruption. For FSQA teams, this makes supplier quality management software a core risk control layer, not an operational nice-to-have.
Regulatory pressure reinforces this reality:
- Food Safety Modernization Act requires documented supplier verification programs
- SQF Edition 9 and BRCGS Issue 9 require structured supplier approval, monitoring, and re-evaluation
- FDA inspection trends consistently show supply chain program deficiencies among the most cited findings

This is where most teams encounter a gap.
Why Generic Quality Control Tools In Manufacturing Fail FSQA Teams
Traditional quality control tools in manufacturing were not designed for food safety compliance. They do not understand:
- Certificates of Analysis validation against specifications
- HACCP linked supplier risks
- FSVP documentation structures
- Audit expectations under GFSI schemes
As a result, FSQA teams revert to spreadsheets, shared drives, and manual follow-ups, which increases risk rather than reducing it.
If your current workflow still depends on manual systems, it reflects the same failure patterns explained here: Why Small Food Manufacturers Fail Food Safety Audits
Who This Guide Is For
This guide is built for:
- QA Managers
- FSQA Directors
- Compliance leaders
Working in food manufacturing, co-packing, and ingredient supply chains, especially teams managing multiple suppliers, SKUs, and audit frameworks.
How This List Was Built
This evaluation focuses on real supplier quality management workflows:
- Alignment with FSMA, SQF, BRCGS, and FSSC
- Depth of supplier document automation
- Implementation speed for mid-sized teams
- Pricing transparency
- Actual automation capability, not just reporting
Most FSQA teams do not realize how fragmented their supplier quality process is until an audit exposes it.
Quick Reference Shortlist
Choosing supplier quality management software is rarely about comparing feature lists. For FSQA teams, the decision usually comes down to one question: which system will actually reduce audit risk and manual workload in the shortest time?
The tools below represent the most relevant supplier quality management tools for food manufacturers in 2026. They were selected based on real-world usability in FSQA environments, not just feature breadth. The focus is on systems that handle supplier documentation, compliance workflows, and COA validation in a way that aligns with how food safety teams actually operate.
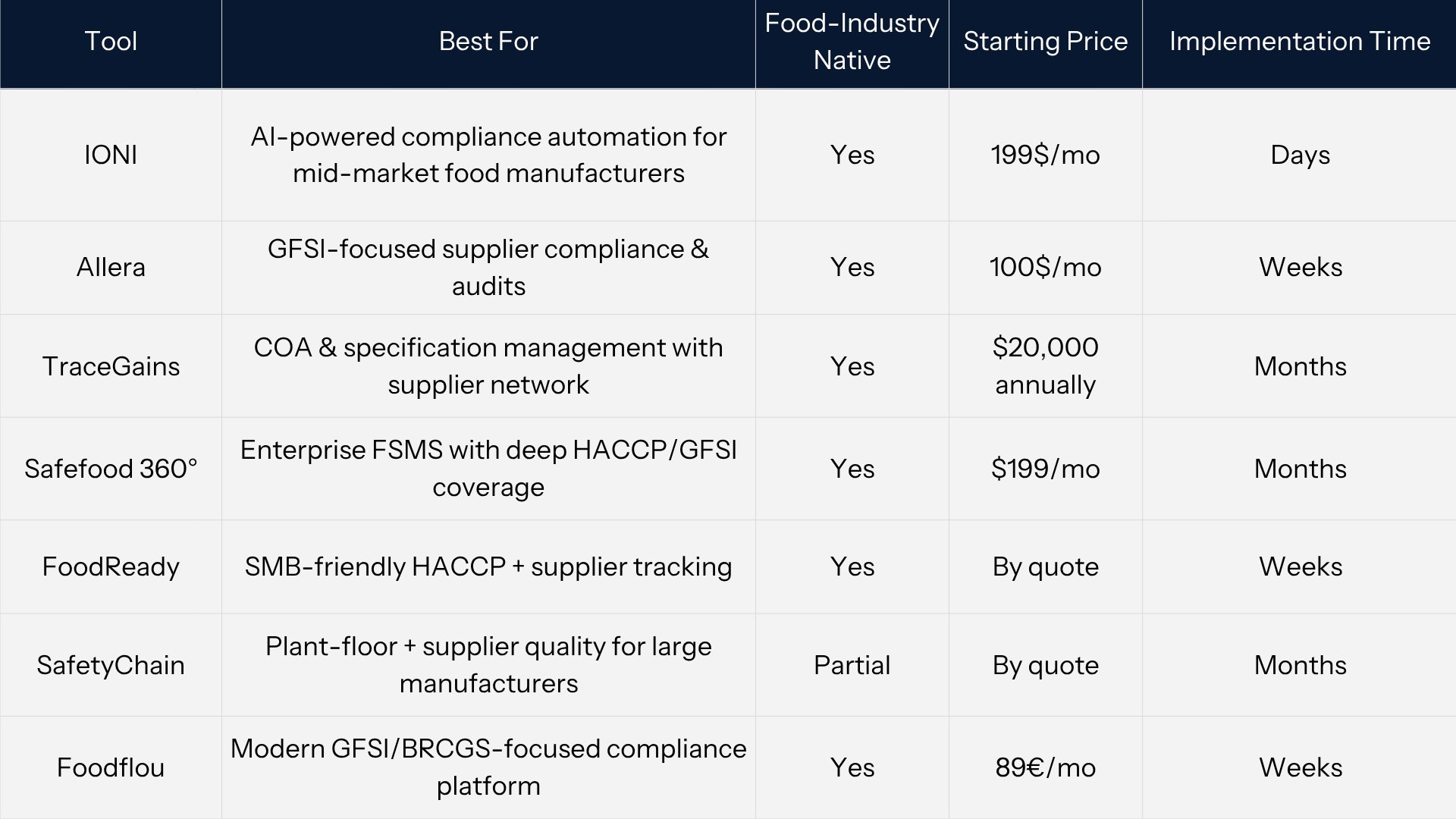
At a high level, the segmentation is clear. Smaller teams tend to prioritize speed and simplicity, while enterprise environments require deeper system coverage and integration. However, the gap between these two groups is where most FSQA teams operate, and it is also where traditional supply chain quality management software often underdelivers.
The most important distinction across these tools is not industry fit, since all are food-focused, but the level of automation. Platforms that still rely on manual document handling will eventually recreate the same bottlenecks teams are trying to eliminate. Systems that automate COA validation, supplier monitoring, and compliance tracking fundamentally change how supplier quality management software performs under real audit pressure.
How We Ranked These Tools
Most lists of supplier quality management software focus on features. FSQA teams do not operate on features. They operate on risk, audit outcomes, and time.
Each platform in this list was evaluated against criteria that directly impact day-to-day supplier quality workflows, audit readiness, and the ability to scale compliance without increasing headcount.
Food Industry Fit
The baseline requirement is simple. The system must understand food safety.
This includes native support for HACCP structures, supplier approval programs, Certificates of Analysis validation, and audit expectations under SQF Edition 9 and BRCGS Issue 9.
Tools that require heavy customization to fit these workflows were ranked lower, regardless of overall functionality.
Supplier Workflow Coverage
Supplier quality management software must cover the full lifecycle:
- Supplier onboarding
- Document collection
- Verification and approval
- Ongoing monitoring and re-evaluation
Fragmented systems that split these steps across modules or external tools introduce risk. In practice, gaps between these stages are where expired certificates, missing COAs, and audit findings occur.

Compliance Depth
The system must align with regulatory and certification requirements without forcing teams to rebuild their processes.
This includes support for Food Safety Modernization Act, FSVP documentation, and GFSI schemes such as FSSC, SQF, and BRCGS. For teams managing multiple frameworks, alignment is not optional. It is operational.
Implementation Speed
Time to value is a critical constraint. FSQA teams cannot afford multi-month implementations while audits continue on fixed schedules. Systems were evaluated based on how quickly a team can move from spreadsheets and shared drives to a functioning supplier quality management workflow.
AI And Automation Capability
This is the primary differentiator in modern supply chain quality management software.
The question is not whether automation exists, but whether it removes real work:
- Automatic document parsing instead of manual review
- COA validation against specifications without human comparison
- Certificate expiration tracking without spreadsheets
- Continuous compliance gap detection instead of periodic checks
Platforms that rely on manual input with digital storage were ranked significantly lower than those that actively validate and interpret data.
Pricing Transparency
Most FSQA teams do not control enterprise-level budgets. Tools were evaluated on whether pricing is understandable and predictable without extended sales cycles, hidden modules, or consulting dependencies. Transparent pricing directly impacts adoption speed and internal approval.
Tool-by-Tool Breakdowns
Most supplier quality management software looks similar at a surface level. Document storage, supplier records, and audit logs. The difference only becomes clear under real FSQA pressure, when teams are managing dozens of suppliers, hundreds of documents, and an upcoming audit.
This section focuses on how each platform performs in actual food manufacturing environments, not just what it claims to do.
#1 IONI - Supplier Compliance Automation System
Website: https://ioni.ai/
What It Does
IONI is not designed as a document repository. It is designed as an execution layer for compliance.
Instead of storing supplier documents, it reads, structures, and continuously validates them against your specifications, HACCP plans, and regulatory requirements. The system effectively replaces manual supplier review workflows with automated validation.
This is a critical distinction. Most supplier quality management tools help you organize documents. IONI helps you decide whether those documents are compliant before they impact production.
Key Features For FSQA Teams
- Automated COA validation against specifications
Every incoming Certificate of Analysis is checked against defined limits before use. This eliminates manual comparison and reduces the risk of non-compliant ingredients entering production.
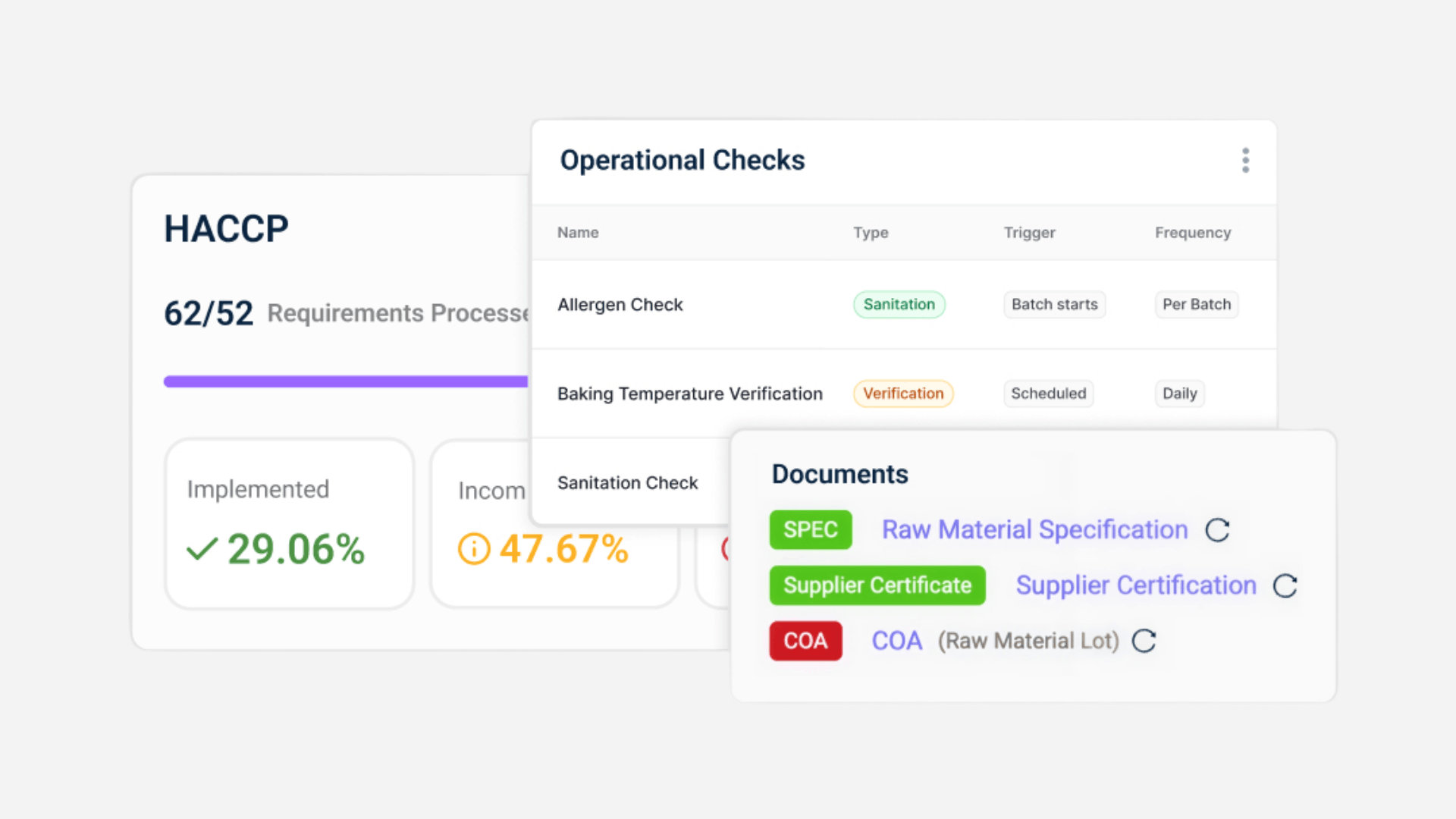
- Supplier document intelligence, not storage
Certificates, specifications, allergen declarations, and approvals are parsed and structured automatically. Missing or inconsistent data is flagged immediately. - Continuous compliance gap detection
The system maps supplier data against SQF Edition 9, BRCGS Issue 9, and Food Safety Modernization Act requirements and highlights gaps before audits.
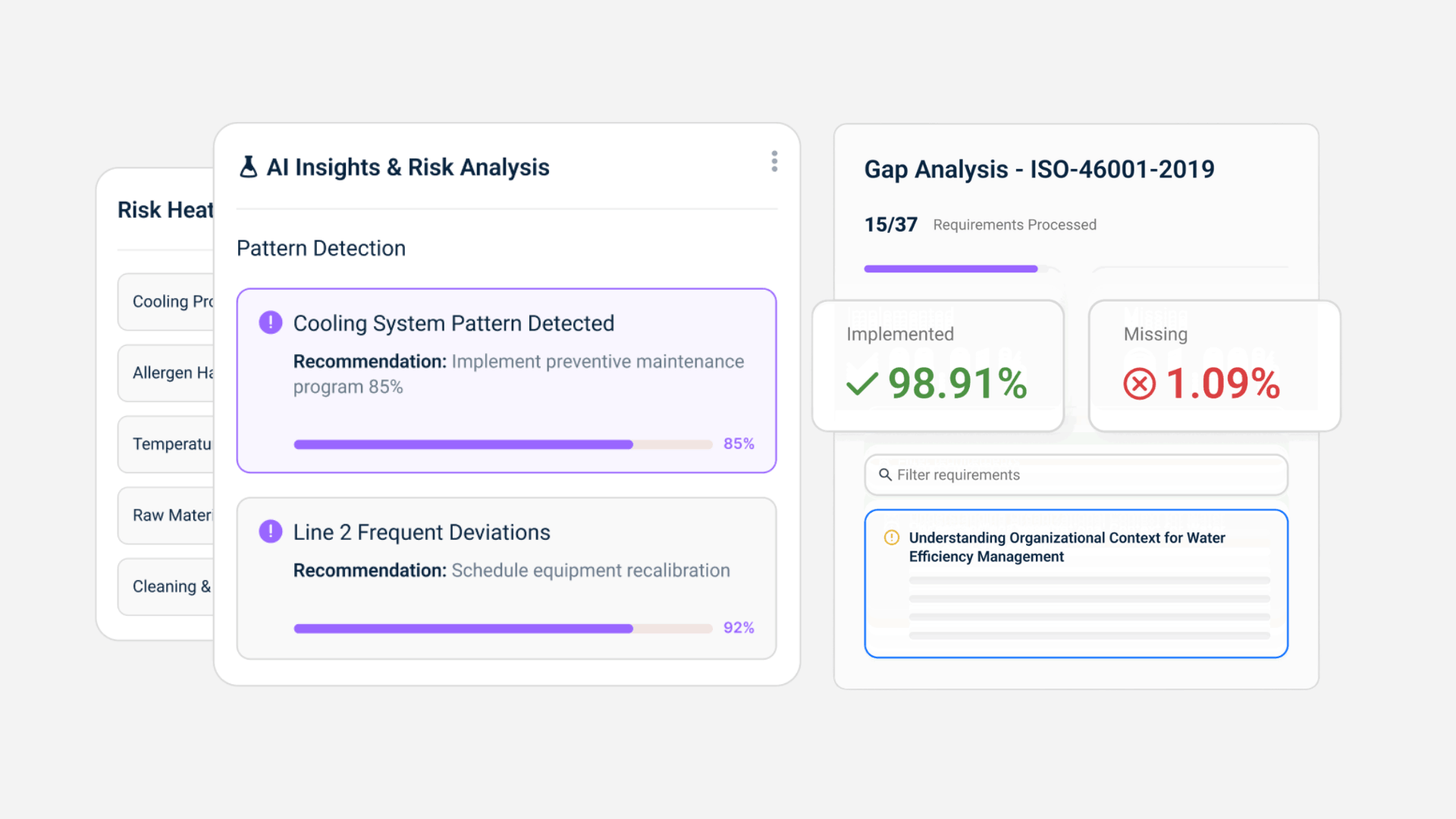
- Real-time supplier risk visibility
Instead of static approval lists, suppliers are dynamically ranked based on compliance status, missing documentation, and historical issues. - Automated audit readiness
Audit evidence is continuously built as the system operates. Instead of preparing for audits, teams operate in a constant state of readiness. - Integrated regulatory intelligence
Regulatory changes are tracked and mapped to your documents and supplier requirements.
- Ingredient-level intelligence and traceability context
Supplier data is connected to ingredient specifications and batch-level risk.
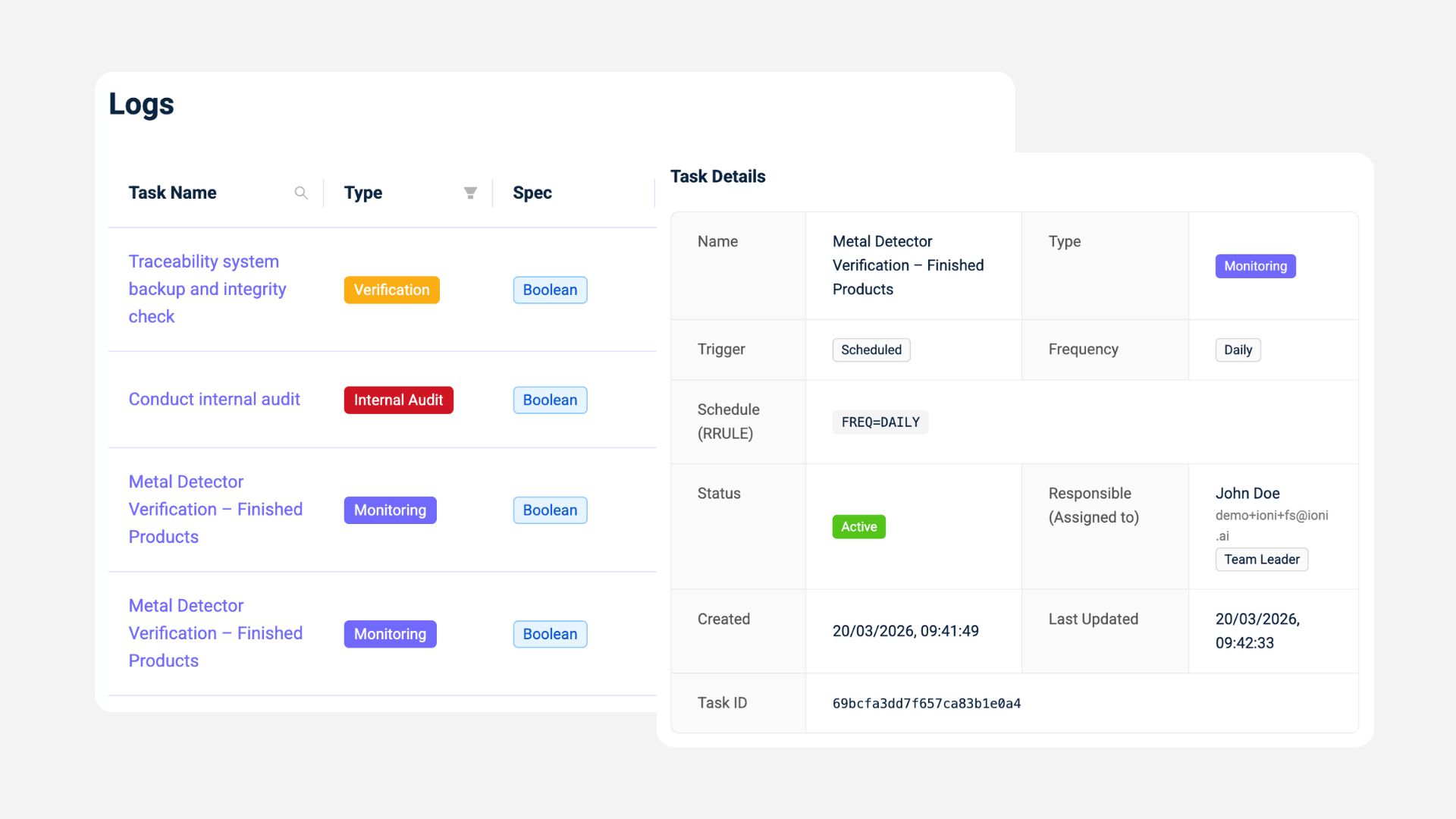
Best For / Focus / Implementation / Pricing
- Best for mid-market food manufacturers scaling supplier quality without increasing QA headcount
- Food focus is native
- Implementation time is measured in days, not weeks or months
- Pricing starts from 199$/mo.
Limitations
- Requires defined specifications to automate validation logic fully
- Less relevant for organizations outside regulated food manufacturing
- Teams expecting a traditional “form-based system” may need to adjust to a more automated workflow model
If your current workflow still relies on manual COA checks, email-based supplier follow-ups, and last-minute audit preparation, it is worth seeing how automation changes that process in practice.
See How IONI Works For Food Safety And Supplier Compliance
#2 Allera - Structured Supplier Quality Management Tools For GFSI
Website: https://www.alleratech.com/
What It Does
Allera provides a structured environment for managing supplier documentation and audit workflows, with strong alignment to GFSI standards.
Key Features
- Supplier document tracking and approval
- Audit workflows and reporting
- Predefined compliance structures
Best For / Focus / Implementation / Pricing
- Best for audit-driven teams preparing for certification
- Implementation typically takes weeks
- Pricing starts from 100$/mo.
Limitations
- Relies on manual document verification
- Limited automation in COA validation and compliance checks
- Workflow-driven rather than intelligence-driven
#3 TraceGains - Supply Chain Quality Management Software With Network Effect
Website: https://tracegains.com/
What It Does
TraceGains combines a large supplier network with compliance and specification management tools, making it a strong option for organizations with complex ingredient sourcing.
Key Features
- Access to an extensive supplier network
- Specification and COA management
- Integrated regulatory data
Best For / Focus / Implementation / Pricing
- Best for large manufacturers with complex supply chains
- Implementation often takes months
- Pricing: specialized modules begin at roughly $20,000 annually
Limitations
- High cost relative to mid-market needs
- Long onboarding cycles
- Complexity can slow down day-to-day FSQA workflows
#4 Safefood 360° - Enterprise Quality Control Tools In Manufacturing
Website: https://safefood360.com/
What It Does
Safefood 360° is a comprehensive food safety management system that includes supplier management as part of a broader compliance framework.
Key Features
- Full HACCP and FSMS coverage
- Supplier approval and monitoring
- Audit and CAPA workflows
Best For / Focus / Implementation / Pricing
- Best for multi-site enterprise operations
- Implementation requires months
- Old listings indicate entry points around $199/mo
Limitations
- Heavy implementation and configuration
- Less flexible for teams needing fast deployment
- Supplier workflows are part of a larger system, not the core focus
#5 FoodReady - Accessible Supplier Quality Management Software For SMBs
Website: https://foodready.ai/
What It Does
FoodReady simplifies compliance for smaller manufacturers by providing structured HACCP and supplier tracking tools.
Key Features
- HACCP plan builder
- Basic supplier document tracking
- Compliance templates
Best For / Focus / Implementation / Pricing
- Best for small teams starting structured compliance
- Implementation takes weeks
- Pricing: no information (by quote)
For a deeper comparison of capabilities and tradeoffs: IONI Vs FoodReady Comparison For Food Manufacturers.
Limitations
- Manual document workflows at scale
- Limited automation in supplier validation
- Less suitable for growing operations with complex supplier bases
#6 SafetyChain - Integrated Manufacturing And Supplier Quality Platform
Website: https://safetychain.com/
What It Does
SafetyChain connects plant floor data with quality and supplier management processes, focusing on large-scale manufacturing environments.
Key Features
- Plant floor data capture and monitoring
- Supplier quality tracking
- Reporting and analytics
Best For / Focus / Implementation / Pricing
- Best for large manufacturers with integrated operations
- Food industry focus is partial
- Implementation requires months
- Pricing: no information (by quote)
Limitations
- Complex deployment
- Supplier quality is not the primary focus
- Limited automation in document validation
#7 Foodflou - Modern Supplier Quality Management Tools For GFSI Workflows
Website: https://www.foodflou.com/
What It Does
Foodflou is a newer platform designed to modernize compliance workflows with a focus on GFSI alignment.
Key Features
- Document management
- Supplier tracking
- Audit workflows
Best For / Focus / Implementation / Pricing
- Best for teams replacing legacy systems
- Implementation takes weeks
- Pricing: starts with 89€/mo
Limitations
- Limited track record compared to established platforms
- Less depth in automation capabilities
- Still evolving in terms of scalability and feature maturity
Across all tools, the pattern is consistent. Most platforms improve organization. A smaller group improves workflow efficiency. Very few fundamentally reduce the manual effort required to manage supplier quality.
That distinction is what defines the next generation of supplier quality management software.
The FSQA Buyer’s Checklist: What Supplier Quality Management Software Must Do
Most evaluations of supplier quality management software fail because they focus on features instead of outcomes. FSQA teams are not buying software to store documents. They are buying a system that reduces supplier risk, prevents audit findings, and eliminates manual workload.
This checklist reframes the evaluation process around capabilities that directly impact food safety, compliance performance, and operational efficiency.
.png)
GFSI Scheme Alignment
The system must natively support the structure and expectations of GFSI schemes, including SQF Edition 9, BRCGS Issue 9, and FSSC 22000.
This includes supplier approval workflows, documentation structures, and audit evidence formats. If the system requires customization to align with these standards, it introduces risk and additional workload.
FSMA Compliance & FSVP
For teams operating in or exporting to the United States, alignment with the Food Safety Modernization Act is mandatory.
The software must support Foreign Supplier Verification Program requirements, including supplier evaluation, verification activities, and documented reassessment. Systems that treat this as generic document storage do not meet regulatory expectations.
For a deeper view on how these frameworks intersect in practice: Food Safety Compliance Across FSMA BRCGS, and SQF.
COA & Specification Management
This is one of the highest impact areas in supplier quality management. The system must not only store Certificates of Analysis but also actively validate them against defined specification limits. Manual comparison at scale is one of the most common failure points in FSQA workflows.
Advanced supplier quality management software should:
- Parse COAs automatically
- Compare results against specifications
- Flag deviations before materials are released
When connected to ingredient-level data, this becomes a core risk prevention layer: Ingredients Intelligence for Food Companies.
Supplier Onboarding & Document Collection
Supplier onboarding must be structured and scalable. The system should allow suppliers to upload required documents directly through a portal, enforce document requirements, and standardize data collection. Manual email-based collection is not sustainable beyond a small number of suppliers.
Certificate Expiration Tracking And Alerts
Expired certifications are a frequent source of audit findings. The system must proactively track expiration dates and trigger alerts before documents lapse. Ideally, this includes automated notifications to both internal teams and suppliers.
Audit Management & CAPA Workflows
Supplier quality management software must support internal and supplier audits, including:
- Audit scheduling and execution
- Non-conformance logging
- Corrective and preventive action tracking
- Verification and closure
The key requirement is traceability. Every issue must be documented, resolved, and verifiable during audits.
Supplier Scorecarding And Performance Visibility
Static supplier approval lists are not sufficient.
The system should provide dynamic supplier scoring based on compliance status, document completeness, historical performance, and risk indicators. This enables FSQA teams to prioritize attention where it matters most.
AI Assisted Compliance Automation
This is the defining capability in modern supply chain quality management software.
The system should reduce manual work by:
- Interpreting supplier documents automatically
- Identifying inconsistencies and missing data
- Mapping supplier information to compliance requirements
- Continuously detecting gaps instead of relying on periodic reviews
This shift from manual verification to automated validation is already reshaping compliance practices across regulated markets: The Role Of AI In Modern SFCR Compliance.
Supplier Quality Management Software: Common Questions From FSQA Teams
Food Safety and Quality Assurance (FSQA) teams are fully responsible for ensuring food products are safe and meet quality standards, acting as both brand protectors and operational partners. Let’s take a look at what questions they may be interested in.
What Is Supplier Quality Management Software?
Supplier quality management software is a system designed to manage supplier approval, documentation, monitoring, and compliance to ensure that all incoming materials meet food safety, regulatory, and specification requirements.
In food manufacturing, this extends beyond record-keeping. It includes active validation of supplier data and continuous monitoring of compliance status.
What Does Supplier Quality Management Software Track In Food Manufacturing?
A complete system tracks all supplier-related compliance data, including:
- Certificates of Analysis and test results
- Supplier certifications and approvals
- Product specifications and limits
- Allergen and ingredient declarations
- Audit results and corrective actions
- Expiration dates and compliance gaps
The depth of tracking determines whether the system supports audits or merely documents activity.
What Is The Difference Between SQM And SRM Software?
Supplier Quality Management (SQM) software focuses on compliance, food safety, and risk control.
At the same time, Supplier Relationship Management (SRM) software focuses on procurement, pricing, and supplier performance from a commercial perspective.
Food manufacturers require SQM capabilities because regulatory compliance cannot be managed through procurement tools.
Should Food Manufacturers Use Food-Specific Supply Chain Quality Management Software?
Yes. Food-specific systems are designed to handle HACCP, COA validation, and regulatory frameworks such as the Food Safety Modernization Act and GFSI schemes.
Generic quality control tools in manufacturing require significant customization and still fail to meet audit expectations, which increases compliance risk.
What Documents Should Supplier Quality Management Software Collect?
A complete supplier file typically includes:
- Certificates of Analysis
- GFSI certifications
- Allergen declarations
- Product specifications
- Supplier questionnaires
- Audit reports
The system must not only collect these documents but also ensure they are current, complete, and compliant.
How Does Supplier Quality Management Software Support GFSI Audits?
During audits, FSQA teams must demonstrate that supplier approval and monitoring are active, not static.
The software supports this by ensuring:
- All required documents are available and up to date
- Records are timestamped and traceable
- Non-conformances are tracked and resolved
- Corrective actions are verified
Teams preparing for certification often follow structured preparation timelines: SQF Certification In 90 Days Guide.
What Is The ROI Of Switching From Spreadsheets To Supplier Quality Management Software?
The ROI is driven by three factors.
First, a reduction in manual workload. FSQA teams spend a significant portion of their time collecting, reviewing, and organizing supplier documents. Automation reduces this effort substantially.
Second, faster audit preparation. Instead of assembling documentation before audits, teams operate in a state of continuous readiness.
Third, risk reduction. Early detection of missing documents, expired certifications, or specification deviations prevents non-conformances and potential recalls.
For most food manufacturers, the return is realized within the first audit cycle due to time savings and reduced compliance risk.
Bottom Line: Which Tool Is Right For Your Team
Selecting the right supplier quality management software depends on where your operation sits in terms of scale, complexity, and compliance maturity.
Small teams that are building their first structured system often prioritize simplicity and cost. Tools like FoodReady provide a practical entry point.
Enterprise manufacturers with multiple facilities, complex operations, and dedicated IT resources typically require systems like Safefood 360° or SafetyChain, where depth and integration matter more than speed.
Organizations managing large supplier networks may benefit from platforms like TraceGains, particularly when supplier connectivity is a priority. However, most FSQA teams fall into a different category. They are managing growing supplier bases, increasing regulatory pressure, and limited internal resources.
In this context, the limitation is not a lack of tools. It is the amount of manual work required to operate them. Traditional supply chain quality management software improves organization, but still depends heavily on human validation. As supplier volume increases, this model does not scale.
IONI approaches supplier quality management differently by automating validation, connecting supplier data to compliance frameworks, and continuously identifying gaps. This shifts supplier quality management from a reactive process to a proactive system.
For FSQA teams that need to scale compliance without scaling headcount, this is the key distinction. The most practical way to evaluate fit is to look at your current supplier workflow. If most of the work involves reviewing documents, chasing updates, and preparing for audits, then automation is not a feature. It is a requirement.
If you are evaluating supplier quality management software and want to understand how this would work with your actual suppliers, specifications, and audit requirements, the most effective next step is to review it in your own context.
Explore AI-Driven Regulatory And Supplier Compliance In Practice









